Which element is an example of a metalloid

Ava Robinson
Published Apr 19, 2026
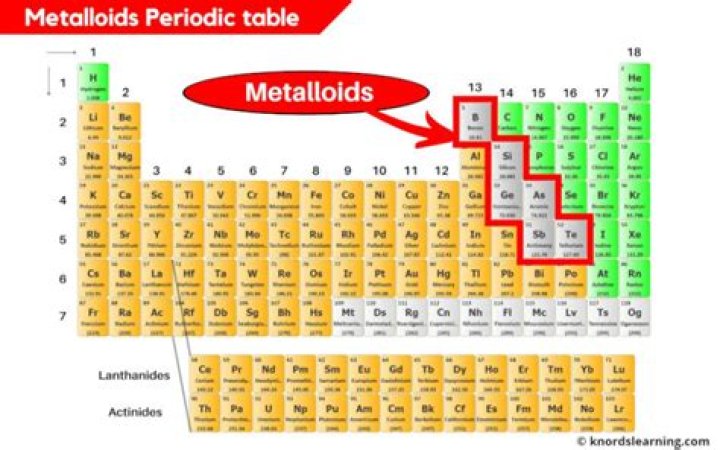
Boron, silicon, germanium, arsenic, antimony, and tellurium are commonly recognised as metalloids. Depending on the author, one or more from selenium, polonium, or astatine are sometimes added to the list.
Which element is a metalloid?
Survey of Elements Commonly Recognized as Metalloids The percentage appearance frequencies of the elements most frequently identified as metalloids are boron (86), silicon (95), germanium (96), arsenic (100), selenium (23), antimony (88), tellurium (98), polonium (49), and astatine (40).
Which element is a metalloid quizlet?
Located in between the metals and the nonmetals on the periodic table. A metalloid is an element that has physical and chemical properties of both metals and nonmetals. The elements boron, silicon, germanium, arsenic, antimony, tellurium, polonium, and astatine are metalloids.
What are 3 metalloids on the periodic table?
They are boron, silicon, arsenic, germanium, antimony, tellurium, polonium, and astatine. There is some disagreement as to some of the elements as metalloids.What is metalloids and examples?
Elements which show some properties of metals and some other properties of nonmetals are called metalloids. Metalloids look like metals but they are brittle like non metals. … They are also called as semi metals. Some important examples of metalloids are as follows : Boron(B), Silicon(Si) and Germanium(Ge).
What are metalloids give two examples Class 8?
- Boron (B)
- Silicon (Si)
- Germanium (Ge)
- Arsenic (As)
- Antimony (Sb)
- Tellurium (Te)
- Polonium (Po)
What are metalloids give any one example?
Boron, silicon, germanium, arsenic, antimony, and tellurium are commonly recognised as metalloids. Depending on the author, one or more from selenium, polonium, or astatine are sometimes added to the list.
Which is not metalloid?
Carbon is not a metalloid and is a non-metal. … The elements which are metalloids, in the periodic table are boron, silicon, arsenic, antimony, polonium, and tellurium. Metalloids are semiconductors.Why is polonium a metalloid?
Discovery date1898Allotropesα-Po, β-Po
Which group contains a metalloid?Groups 13–16 of the periodic table contain one or more metalloids, in addition to metals, nonmetals, or both. Group 13 is called the boron group, and boron is the only metalloid in this group. The other group 13 elements are metals. Group 14 is called the carbon group.
Article first time published onAre metalloids semiconductors?
A number of the metalloids are semiconductors as they have intermediate conductivity which is temperature dependent. The ability of the metalloids to conduct electricity and heat is far better than the nonmetals, for example, diamond , which are insulators.
Which properties do metalloids share with metals?
- An appearance that is similar to metals.
- They are less conductive than metal.
- They are more brittle than metals.
- Metalloids have nonmetallic chemical properties in general.
What are metalloids give two examples Brainly?
Thus, the metalloid is an element intermediate in characteristics between the typical metals as well as nonmetals. Illustrations of Metalloids are Boron (symbol B), Silicon (symbol Si), Germanium (symbol Ge), Arsenic (symbol As), etc.
What is the most common metalloid?
Silicon is the “big sister” to carbon, and shares many of the same chemical properties. For this reason, silicon is known as a chemical analogue to carbon. But unlike carbon, silicon a metalloid — in fact, it’s the most common metalloid on earth.
What are metalloids give Example Class 9?
Metalloids are elements which have properties intermediate between those of metals and non-metals. Examples are silicon, germanium etc. burning of paper and wood.
Is an example of metalloid *?
Boron, silicon, germanium, arsenic, antimony, tellurium, and polonium are metalloids.
What are metalloids give Example Class 11?
Elements which show properties of both metals and non-metals are called metalloids. For example: Silicon, Germanium.
What are metalloids short answer?
a nonmetal that in combination with a metal forms an alloy. an element that has both metallic and nonmetallic properties, as arsenic, silicon, or boron.
What is a metalloid Class 10?
The elements which display properties of both metals and non-metals are classified as metalloids. In the Periodic Table, they separate metals from the non-metals. For eg. Silicon, Arsenic etc. webew7 and 63 more users found this answer helpful.
What are metalloids for Class 8?
Metalloids are the smallest class of elements. (The other two classes of elements are metals and nonmetals). There are just six metalloids. In addition to silicon, they include boron, germanium, arsenic, antimony, and tellurium.
What are metalloids Brainly?
A metalloid is an element that has properties that are intermediate between those of metals and nonmetals. Metalloids can also be called semimetals. On the periodic table, the elements colored yellow, which generally border the stair-step line, are considered to be metalloids.
Is bismuth a metalloid?
Traditionally they include boron from group 3A, silicon and germanium in group 4A, aresnic and antimony in group 5A and tellurium from group 6A, although sometimes selenium, astatine, polonium and even bismuth have also been considered as metalloids. Typically metalloids are brittle and show a semi-metallic luster.
Is Tungsten a metalloid?
Answer: Tungsten is a metal . It is very heavy with a density of around 19.3 gms /cm^3 , which is more than 2.5 times that of the steel.
Is tellurium a metalloid?
Tellurium is a chemical element with symbol Te and atomic number 52. Classified as a metalloid, Tellurium is a solid at room temperature.
Is bromine a metalloid?
Bromine is the third halogen, being a nonmetal in group 17 of the periodic table.
Is boron a metalloid?
The metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium. These elements look metallic; however, they do not conduct electricity as well as metals so they are semiconductors. … Their chemical behavior falls between that of metals and nonmetals.
Is the liquid metal?
The only stable liquid elemental metal at room temperature is mercury (Hg), which is molten above −38.8 °C (234.3 K, −37.9 °F). … Alloys can be liquid if they form a eutectic, meaning that the alloy’s melting point is lower than any of the alloy’s constituent metals.
Why is carbon a metalloid?
Carbon is a non-metal. It belongs to the fourteenth group or IV A group in the modern periodical table. The elements of this group have four electrons in the valence shell. … To get the octet in its outer shell it has to gain four more electrons to form C4-.
Are group 5 elements metals or nonmetals?
Group 5A (or VA) of the periodic table are the pnictogens: the nonmetals nitrogen (N), and phosphorus (P), the metalloids arsenic (As) and antimony (Sb), and the metal bismuth (Bi).
Is a metal nonmetal or metalloid?
7B—8B—
What element is a metalloid and semiconductor?
The metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium. These elements look metallic; however, they do not conduct electricity as well as metals so they are semiconductors.