Which caspase is the initiator of apoptosis

Olivia House
Published Apr 21, 2026
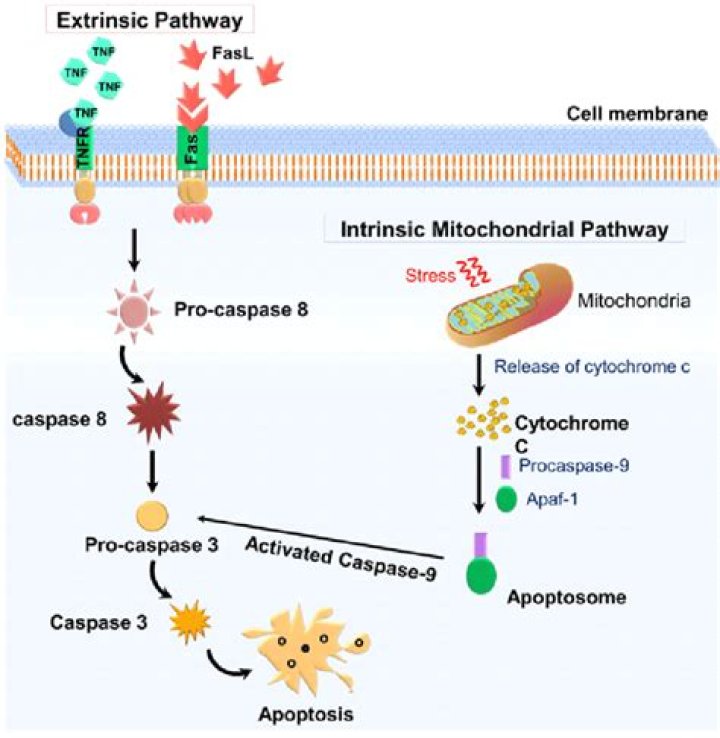
CED-3 in C. elegans is both the initiator and the effector caspase. The executioners of apoptosis are caspases, a family of conserved cysteine proteases that usually cleave after an aspartate residue in their substrates (Thornberry and Lazebnik 1998).
Is caspase 1 an initiator caspase?
Mammalian caspases can be subdivided into three functional groups: initiator caspases (caspase 2, 8, 9, and 10), executioner caspases (caspase 3, 6, and 7), and inflammatory caspases (caspase 1, 4, 5, 11, and 12)3.
Is caspase-8 an initiator caspase?
Caspase-8 (Casp8), which was first identified in humans, functions as an initiator caspase in the apoptotic signaling mediated by cell-surface death receptors.
Is caspase-9 an initiator caspase?
In most known forms of intrinsic apoptosis, caspase-9 and caspase-3/7 are the initiator and the downstream effector caspases, respectively.Is caspase 1 involved in apoptosis?
Caspases are a family of cysteine proteases that mediate regulated cell death, including apoptosis and pyroptosis1.
What is the role of caspase 3 in apoptosis?
Caspase-3 is known as an executioner caspase in apoptosis because of its role in coordinating the destruction of cellular structures such as DNA fragmentation or degradation of cytoskeletal proteins (1).
Are caspases secreted?
Inflammatory protease, caspase-1, is secreted from monocytes in a stably active form in response to inflammasome activation (INM3P. 441)
How does caspase-9 cause apoptosis?
Active caspase-9 works as an initiating caspase by cleaving, thus activating downstream executioner caspases, initiating apoptosis. Once activated, caspase-9 goes on to cleave caspase-3, -6, and -7, initiating the caspase cascade as they cleave several other cellular targets.How does caspase-3 and 7 cause apoptosis?
Caspase-3 and caspase-7 are both activated universally during apoptosis, irrespective of the specific death-initiating stimulus, and both proteases are widely considered to coordinate the demolition phase of apoptosis by cleaving a diverse array of protein substrates (1, 2).
Where is caspase-3 activated?Activation. Caspase-3 is activated in the apoptotic cell both by extrinsic (death ligand) and intrinsic (mitochondrial) pathways. The zymogen feature of caspase-3 is necessary because if unregulated, caspase activity would kill cells indiscriminately.
Article first time published onWhat are initiator and effector caspases?
An initiator caspase invariably contains an extended N-terminal prodomain (>90 amino acids) important for its function, whereas an effector caspase frequently contains 20–30 residues in its prodomain sequence.
Is caspase-8 an effector caspase?
Caspase-8 is an effector in apoptotic death of dopaminergic neurons in Parkinson’s disease, but pathway inhibition results in neuronal necrosis. J Neurosci.
Is caspase-8 extrinsic?
Caspase-8 (CASP8) is a cysteine protease that plays a pivotal role in the extrinsic apoptotic signaling pathway via death receptors. The kinetics, dynamics, and selectivity with which the pathway transmits apoptotic signals to downstream molecules upon CASP8 activation are not fully understood.
How does caspase-1 cause apoptosis?
Caspase-1 activated in inflammasomes triggers a programmed necrosis called pyroptosis, which is mediated by gasdermin D (GSDMD). … Chemical dimerization of caspase-1 induces pyroptosis in GSDMD-sufficient cells, but apoptosis in GSDMD-deficient cells.
What is the role of caspase-1?
Caspase-1 is a cysteine protease that converts the inactive proform of IL-1β to the active inflammatory cytokine and hence represents an attractive target for the modulation of the effects of IL-1β (12,13).
What secretes IL1?
IL-1 is produced predominantly by macrophages and macrophage-like cells but also by endothelial and epithelial cells. … During development, IL-1 production by fetal macrophages in response to LPS up-regulates G-CSF production by monocytes from the bone marrow and liver of human fetuses.
What is caspase activation?
Activation of caspases ensures that the cellular components are degraded in a controlled manner, carrying out cell death with minimal effect on surrounding tissues. … Caspases have other identified roles in programmed cell death such as pyroptosis and necroptosis.
Which amino acids are associated with caspase?
Caspases are a family of cysteine proteases that cleave proteins following aspartic acid residues. These proteases exist in a hierarchy with upstream caspases 2, 8, 9, and 10 and downstream caspases 3, 6, and 7.
What initiates apoptosis?
Apoptosis is mediated by proteolytic enzymes called caspases, which trigger cell death by cleaving specific proteins in the cytoplasm and nucleus. Caspases exist in all cells as inactive precursors, or procaspases, which are usually activated by cleavage by other caspases, producing a proteolytic caspase cascade.
How does caspase help apoptosis?
Caspases are a family of cysteine proteases that serve as primary effectors during apoptosis to proteolytically dismantle most cellular structures, including the cytoskeleton, cell junctions, mitochondria, endoplasmic reticulum, Golgi, and the nucleus (Taylor et al., 2008).
What is the function of caspase-8?
Caspase-8 is a critical molecule as its absence leads to death of mice in utero. Upon activation, its main function is to promote apoptosis and, in thymus, apoptosis of negative selection is critical to eliminate autoaggressive T-cell clones that, if not eliminated, could contribute to develop autoimmune diseases.
Which caspase play vital role in final phase of apoptosis?
Recent work, reviewed here, has revealed that caspase-3 is important for cell death in a remarkable tissue-, cell type- or death stimulus-specific manner, and is essential for some of the characteristic changes in cell morphology and certain biochemical events associated with the execution and completion of apoptosis.
What does caspase 7 do in apoptosis?
However both of these effector caspases have distinct functions during apoptosis. Recent studies have indicated caspase-3 is needed for efficient cell killing and can also block ROS production (3). Caspase-7, on the other hand, is responsible for ROS production and aids in cell detachment during apoptosis (3).
What is caspase Glo?
The Caspase-Glo® 3/7 Assay(a,b) is a homogeneous, luminescent assay that measures caspase-3 and -7 activities. … The Caspase-Glo® 3/7 Assay is designed for use with multiwell-plate formats, making it ideal for automated high- throughput screening of caspase activity or apoptosis.
Is caspase 3 a marker of apoptosis?
Apoptotic cell death is fundamental cell process that utilizes the cell death receptor family signaling network. Caspases are activated by a cleavage that occurs adjacent to an aspartate; this cleavage liberates the individual units and allows formation of an a2b2 tetramer. …
Does cytochrome c activate caspase-9?
Caspase-9 is activated in a cytochrome c-independent manner early during TNFalpha-induced apoptosis in murine cells. Cell Death Differ.
What is cleaved caspase-8?
In the context of extrinsic apoptosis, caspase-8 is activated by dimerization inside a death receptor complex, cleaved by auto-proteolysis and subsequently released into the cytosol. This fully processed form of caspase-8 is thought to cleave its substrates BID and caspase-3.
What is activated caspase 3?
Caspase-3 is a cysteine–aspartic acid protease that cleaves cellular targets and executes cell death. Our current understanding is caspase-3 is activated by the cleavage of the interdomain linker and then subsequent cleavage of the N-terminal prodomain.
Where are caspases found?
Pro-caspases-7 and -8 were found only in the cytosolic fraction. In apoptotic cells, caspases-3, -8 and -9 were present in the cytosolic fraction, whereas caspases-3 and -9 were also found in the mitochondrial fraction and caspase-7 in the microsomal fraction.
What is the role of caspases in apoptosis Mcq?
Explanation: Caspases are the effectors of apoptosis. IAP binds to the caspases thus preventing apoptosis from taking place. Smac and Diablo bind to IAP thus inhibiting it so they are also pro-apoptotic.
Is caspase 3 an effector caspase?
Caspase-3, caspase-6, and caspase-7 are effector caspases that execute apoptosis. Activated caspase-3 and caspase-7 can cleave multiple structural and regulatory proteins, which are critical for cell survival and maintenance (Boyce et al., 2004; Crawford & Wells, 2011).