Where are glycosidic linkages found

Andrew Mitchell
Published Apr 21, 2026
Glycosidic linkages are commonly found in carbohydrates, such as sugars and starches.
Where are glycosidic bonds found?
What Is a Glycosidic Bond? They are in the sugar that you eat, the trunks of trees, the hard exoskeleton of lobsters, and even in your DNA. Glycosidic bonds are important for the structure of all of these substances and many others.
Where is a glycosidic bond located in DNA?
A glycosidic bond exists in the DNA molecule between sugar and nitrogen base. The glycosidic bond is formed by the nitrogen-carbon linkage between the 9′ nitrogen of purine bases or 1′ nitrogen of pyrimidine bases and the 1′ carbon of the sugar group. Sugar present in DNA is deoxyribose.
Where is a glycosidic linkage found in a disaccharide?
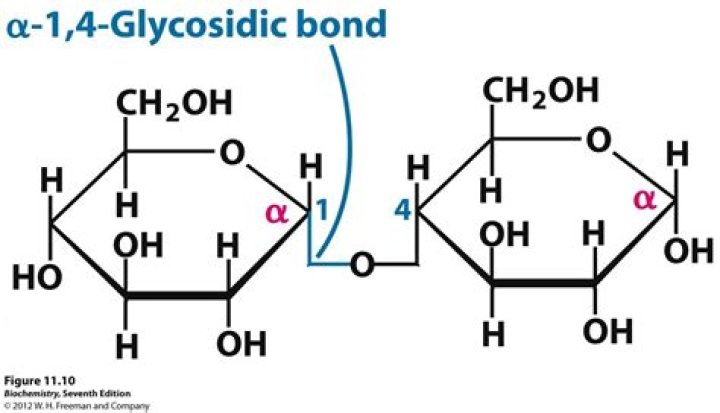
Disaccharides consist of two monosaccharide units, linked by glycosidic bonds in the α or β orientation.Is glycosidic linkage present in glucose?
In cellulose, glucose monomers are linked in unbranched chains by β 1-4 glycosidic linkages.
Is an ester bond covalent?
Ester Bond It is a covalent bond that is essential in various types of lipids. An ester bond or ester linkage is formed between an acid and an alcohol.
Does glycogen have alpha or beta linkages?
Glycogen is found in animals, and it is branched like amylopectin. It is formed by mostly alpha 1,4 glycosidic linkages but branching occurs more frequently than in amylopectin as alpha 1,6 glycosidic linkages occur about every ten units. … Cellulose is made of repeating beta 1,4-glycosidic bonds.
Which glycosidic linkage is present in maltose?
β− glycosidic linkage is present in maltose.How do you identify disaccharide linkages?
identify disaccharides as compounds consisting of two monosaccharide units joined by a glycoside link between the C1 of one sugar and one of the hydroxyl groups of a second sugar.
What is amylose composed of?Amylose is a polysaccharide made of α-D-glucose units, bonded to each other through α(1→4) glycosidic bonds. It is one of the two components of starch, making up approximately 20-30%.
Article first time published onWhat type of bonds are found in DNA backbone?
Bases are held together by hydrogen bonds, and the DNA backbone is held together by phosphodiester bonds.
Which of the following bond is present in DNA?
Covalent bonds occur within each linear strand and strongly bond the bases, sugars, and phosphate groups (both within each component and between components). Hydrogen bonds occur between the two strands and involve a base from one strand with a base from the second in complementary pairing.
Which type of linkage is a glycosidic linkage?
A glycosidic bond or glycosidic linkage is a type of covalent bond that joins a carbohydrate (sugar) molecule to another group, which may or may not be another carbohydrate.
Which of the following linkage is found in sucrose?
1−2 glycosidic linkage.
What is the linkage between glucose and fructose?
In sucrose, the components glucose and fructose are linked via an acetal bond between C1 on the glucosyl subunit and C2 on the fructosyl unit. The bond is called a glycosidic linkage. Glucose exists predominantly as two isomeric “pyranoses” (α and β), but only one of these forms links to the fructose.
Which of the following linkages are seen in cellulose?
In cellulose, the glucose units are linked by β- glycosidic linkage.
What type of linkages are found in glycogen?
Glycogen is a polysaccharide that consists entirely of glucose molecules that are linked by two types of bonds – the alpha-1,4-glycosidic bond and the alpha-1,6-glycosidic bond. The alpha-1,4-glycosidic bond is the more common bond and it gives glycogen a helical structure that is suitable for energy storage.
Does starch have alpha or beta linkages?
Starch is formed from alpha glucose, while cellulose is made of beta glucose. The difference in the linkages lends to differences in 3-D structure and function. Starch can be straight or branched and is used as energy storage for plants because it can form compact structures and is easily broken down.
What are beta linkages?
When a compound with beta confirmation links with another compound with beta confirmation, the linkage is beta linkage. Example Latose ( beta D-galactose linked to beta D-glucose with beta 1→4 linkage) 3. When a compound with alpha and beta both compounds linked together, such a linkage is called alpha- beta linkage.
Where are esters found?
Esters are ubiquitous. Most naturally occurring fats and oils are the fatty acid esters of glycerol. Esters with low molecular weight are commonly used as fragrances and found in essential oils and pheromones.
Why are ester linkages important?
Ester linkages are key components of molecules called lipids. In our bodies, lipids form lipid bilayers, which compose cell membranes and other organelles within the cell. They’re able to do this because of their ability to be both hydrophilic and hydrophobic.
What do ester linkages connect?
In an ester molecule, the bond connecting the atom doubly bonded to oxygen and the oxygen atom bearing the alkyl or aryl group is called the ester bond or, in biochemistry, ester linkage.
Are disaccharides and oligosaccharides same?
Oligosaccharides contain glycosidic linkages (acetal or ketal) that release two or more monosaccharide units upon hydrolysis. … Disaccharides are oligosaccharides that contain two monosaccharide units. Polysaccharides contain a large number of monosaccharide units bonded to each other by a series of glycosidic bonds.
What is the function of disaccharides?
In your body, a disaccharide function is to provide your body with a quick source of energy. Because they’re only made up of two sugar molecules, they’re easily broken down by enzymes in your digestive system into their respective monosaccharides and then absorbed into your bloodstream.
What are food examples of disaccharides?
- Derived from sugar cane and sugar beet.
- Table sugar, manufactured foods, such as cakes, cookies, and dark chocolate.
- Sweet root vegetables such as beetroot and carrots.
Which type of linkage is present in starch?
4.1 Starch. Starch consists of two types of polysaccharides: amylose and amylopectin. Amylose is a straight linear chain of glucose molecules linked by α-l,4 glycosidic linkages as shown in Fig. 2.19, in the same manner as in the disaccharide maltose.
Where is amylopectin found?
Amylopectin is a polysaccharide that is found in a molecule of starch. It is composed of multiple glucose units and has a variable structure. More than 80% of amylopectin is found in a starch molecule. The presence of amylopectin can be determined using the iodine test.
What type of linkage is there in amylopectin?
Amylopectin is composed of linear chains of α-(1,4)-D-glucose residues connected through α-(1,6) linkages (5–6%).
Where is amylose found in plants?
It is typically produced in plastids of leaves, seeds and storage organs as semicrystalline insoluble granules. Amylopectin is the major polymer in starch, and has α-1,4-linked glucose chains (degree of polymerisation (DP) < 100) joined by α-1,6-linked branch points (Fig.
Which bond is not found in the sugars that make up DNA?
Hydrogen bonds are not chemical bonds. They can be easily disrupted. This permits the DNA strands to separate for transcription (copying DNA to RNA) and replication (copying DNA to DNA). In our simple model, the entire base pair structure is represented by the single blue rod.
What types of bonds are involved in stabilizing the DNA backbone and what types of bonds are found between complementary base pairs?
The base pairs are stabilized by hydrogen bonds; adenine and thymine form two hydrogen bonds between them, whereas cytosine and guanine form three hydrogen bonds between them. Figure 7. Hydrogen bonds form between complementary nitrogenous bases on the interior of DNA.