What is the purpose of doing an experiment in a buffer solution

Nathan Sanders
Published Apr 18, 2026
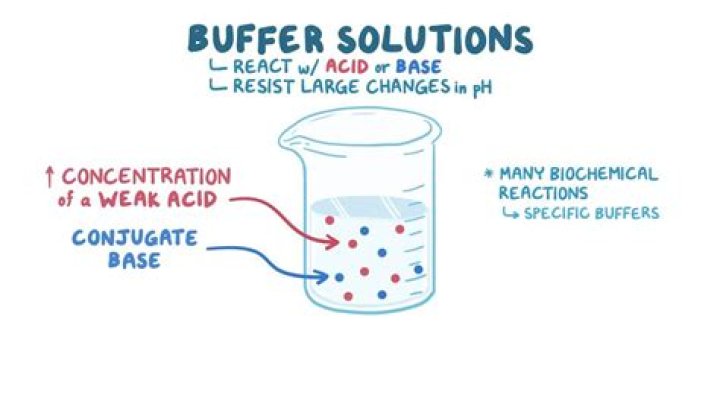
Buffer, as we have defined, is a mixture of a conjugate acid-base pair that can resist changes in pH when small volumes of strong acids or bases are added. When a strong base is added, the acid present in the buffer neutralizes the hydroxide ions (OH -start superscript, start text, negative, end text, end superscript).
What is the purpose of a buffer experiment?
A buffer solution is a distinct solution used in experiments that need to work in a certain pH range. The buffer solution has the capability to prevent sudden changes in the pH of the solution. It is an important solution, especially in working biological samples where there is an optimum pH for the reaction.
How does a buffer help a solution maintain pH quizlet?
a substance that tends to oppose changes in pH of a solution by removing or replacing hydrogen ions; in body fluids, buffers maintain blood pH within normal limits (7.35-7.45).
What is the purpose of buffer in gel electrophoresis?
High-quality buffers are an important part of electrophoresis. They allow a current to be carried through the sample while resisting pH changes in the overall solution. The choice of buffer depends on the isoelectric point of the sample being analyzed.What should be considered in making a buffer solution?
Buffers. A buffer is an aqueous solution containing a weak acid and its conjugate base or a weak base and its conjugate acid. A buffer’s pH changes very little when a small amount of strong acid or base is added to it.
What is the pH of a buffer solution?
A buffer is an aqueous solution designed to maintain a constant pH, even when exposed to small amounts of acids or bases. Whether acidic (pH < 7) or basic (pH > 7), a buffer solution consists of a weak acid or base mixed with the salt of its conjugate base or acid, respectively.
How are buffer solutions prepared?
Buffers can either be prepared by mixing a weak acid with its conjugate base or a weak base with its conjugate acid. For example, phosphate buffer, a commonly used buffer in research labs, consists of a weak base (HPO42-) and its conjugate acid (H2PO4–). Its pH is usually maintained at 7.4.
What is the purpose of a buffer solution quizlet?
The function of a buffer is to resist changes in the pH of a solution when acid (HCl) or base (NaOH) (small amount) is added.What are the two purposes for the buffer in the electrophoresis box?
In gel electrophoresis, the buffer provides ions that carry a current through the gel, and to maintain a constant pH. There are a variety of buffers, and one of the most common for DNA separation is TBE buffer. TBE buffer is a buffer solution containing a mixture of Tris base, boric acid and EDTA.
How do buffer works to maintain the pH of a solution?Buffers work by neutralizing any added acid (H+ ions) or base (OH- ions) to maintain the moderate pH, making them a weaker acid or base. … Now, because all the extra H+ ions are locked up and have formed a weaker acid, NH4+, thus the pH of the system does not change significantly.
Article first time published onWhat is the purpose of a biological buffer quizlet?
Buffers prevent even the slightest changes in pH that can inhibit important biological molecules such as enzymes.
What is buffer action?
Buffer action in general is defined as the ability of the buffer solution to resist the changes in pH value when a small amount of an acid or a base is added to it.
What is buffer solution and types of buffer solution?
A buffer solution is chemical solution which resists change to its pH or acidity. It is a solution in water of a mixture of a weak acid or base and its salt. … Buffer solutions may be of two types: acidic and basic. Acidic : A solution of mixture of weak acid and a salt of this acid with a strong base.
What makes a good pH buffer?
Buffers should have a pKa between 6.0 and 8.0 because the optimal pH for most biological reactions rests in this range. Buffers should have high water solubility and minimum solubility in organic solvents so it remains in the aqueous medium of the biological system. Buffers should not permeate cell membranes.
What will happen to the pH of the buffer if the concentration of the buffer is halved?
pH goes up because there is less total acid in the solution.
What factors affect buffer capacity?
Buffer capacity is the measure of a buffer’s ability to resist pH change. This ability depends on the concentration of the buffer components, meaning the acid and its conjugate base. A higher buffer concentration has a greater buffer capacity.
How would you describe a buffer solution quizlet?
A buffer solution is a system that does not change significantly if small amounts off acid or base are added to it. pH only changes a small amount as the buffer works.
How do buffers work quizlet?
A buffer is a chemical system that resists changes in pH by neutralizing added acid or base. … When base is added to the system, the weak acid neutralizes it, generating more of the conjugate base. When acid is added to the system, the base neutralizes the acid, generating more of the conjugate acid.
Does pH buffer increase pH?
Buffer increases the pH and the Total Alkalinity. To raise only the pH Pool Pro pH Increaser is used. Be careful as it raises the pH very quickly so only small doses are recommended, then test again.
Why do buffers work best when pH pKa?
A buffer works best when there is the same amount of weak acid/base and its conjugate. If you look at the Henderson Hasselbalch equation, and set the concentration of the weak acid/base equal to each other, pH=pKa.
Why is pH so important to biological systems?
Because all biological processes are dependent on pH, cells and organisms must maintain a specific and constant pH in order to keep their enzymes in the optimum state of protonation. It provides the buffering effect in intracellular fluid, and is important in urine. …
Do buffers keep the pH within normal limits?
Buffers usually consist of a weak acid and its conjugate base; this enables them to readily absorb excess H+ or OH–, keeping the system’s pH within a narrow range.
What happens to pH as acidity decreases?
When an acidic solution is diluted with water the concentration of H + ions decreases and the pH of the solution increases towards 7. … This causes the pH of the alkali to fall towards 7, making the solution less alkaline as more water is added.