What is anode simple

Andrew Mitchell
Published Apr 19, 2026
An anode is the electrode in a polarized electrical device through which current flows in from an outside circuit. … Cathodes get their name from cations (positively charged ions) and anodes from anions (negatively charged ions). In a device that consumes electricity, the anode is the charged negative electrode.
What is a anode simple definition?
Definition of anode 1 : the electrode of an electrochemical cell at which oxidation occurs: such as. a : the positive terminal of an electrolytic cell. b : the negative terminal of a galvanic cell.
What is in an anode?
The anode is the positively charged electrode. The anode attracts electrons or anions. The anode may be a source of positive charge or an electron acceptor.
What is cathode and anode in simple words?
The Anode is the negative or reducing electrode that releases electrons to the external circuit and oxidizes during and electrochemical reaction. The Cathode is the positive or oxidizing electrode that acquires electrons from the external circuit and is reduced during the electrochemical reaction.What is anode and example?
Jun 19, 2014. An anode is any electrode where oxidation takes place. A simple example is electrolysis of water. A positively charged platinum electrode where H2 gas is oxidized to H+ ions is the anode.
What is anode class 10th?
The anode is the electrode where electricity moves into. The cathode is the electrode where electricity is given out or flows out of. The anode is usually the positive side. A cathode is a negative side.
What is cathode simple definition?
Definition of cathode 1 : the electrode of an electrochemical cell at which reduction occurs: a : the negative terminal of an electrolytic cell. b : the positive terminal of a galvanic cell.
What do you mean by cathode ray?
cathode ray, stream of electrons leaving the negative electrode (cathode) in a discharge tube containing a gas at low pressure, or electrons emitted by a heated filament in certain electron tubes.What is anode in electrolysis?
An anode is an electrode through which the conventional current enters into a polarized electrical device. … In both a galvanic cell and an electrolytic cell, the anode is the electrode at which the oxidation reaction occurs. In an electrolytic cell, the anode is the wire or plate having excess positive charge.
Is anode +ve or?anode, the terminal or electrode from which electrons leave a system. In a battery or other source of direct current the anode is the negative terminal, but in a passive load it is the positive terminal.
Article first time published onWhy is the anode positive?
In an electrolytic cell(one used for electrolysis), the anode is positively charged. This is because the electrode connected to the positive terminal of the battery is where the negatively charged ions go to lose electrons, that is, to get oxidized.
How do you make an anode?
Grind a mixture of manganese dioxide, potassium hydroxide and graphite into a fine powder and press it into tablets. These tablets will then form the cathode of an alkaline battery. Use a gel that consists primarily of zinc powder for the anode of the battery.
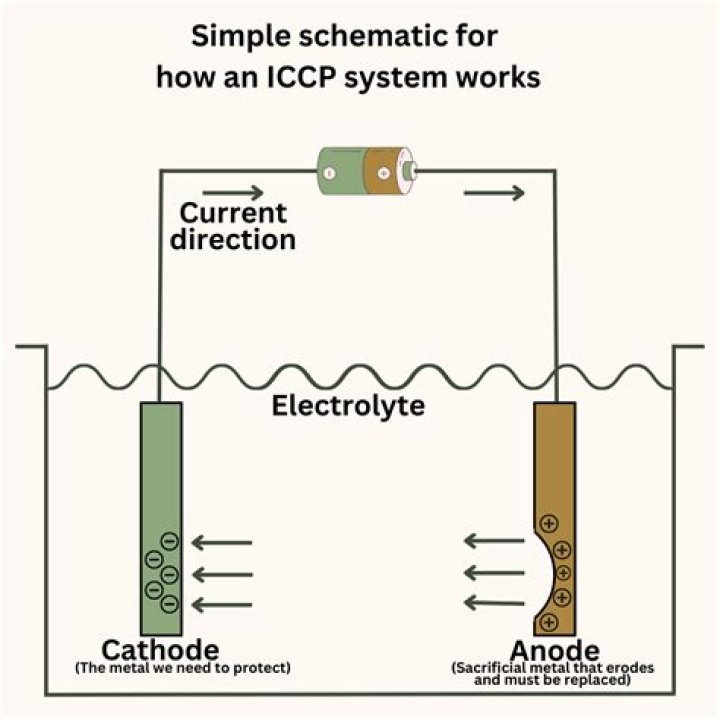
What is an anode in corrosion?
Anode – The electrode where galvanic reaction(s) generate electrons – negative ions are discharged and positive ions are formed. Corrosion occurs at the anode. Cathode – The electrode that receives electrons – positive ions are discharged, negative ions are formed.
What is the difference between cathode and anode?
A cathode is an electrode where the electricity flows out of or given out. An anode is an electrode where the electricity moves into. A cathode is a negative sided electrode. An anode is a positive sided electrode.
What is a positive ion?
Ions are invisible charged particles in the air – either molecules or atoms, which bear an electric charge. … To put it simply, positive ions are molecules that have lost one or more electrons whereas negative ions are actually oxygen atoms with extra-negatively-charged electrons.
How does a cathode work?
How Cathode Ray Tubes Work. … The cathode is a negatively-charged conductor, and the anode is a positively-charged conductor. Electrons, which have a negative charge, flow off the cathode and are attracted towards the anode. A small hole in the anode allows some electrons to pass through it, creating a beam of electrons.
What is cell voltage?
The cell voltage is a quantitative measure of the driving force of the cell reaction. … Hint: In an electrochemical cell, the difference in the electrical potential between two electrodes is known as the cell voltage. If the cell voltage increases the tendency of the cell reaction to occur increases.
How do you remember the anode and cathode?
In chemistry, a cathode is the electrode of an electrochemical cell at which reduction occurs; a useful mnemonic to remember this is AnOx RedCat (Oxidation at the Anode = Reduction at the Cathode). Another mnemonic is to note the cathode has a ‘c’, as does ‘reduction’. Hence, reduction at the cathode.
Why is cathode negative and cation positive?
1: An electrolytic cell. The battery pumps electrons away from the anode (making it positive) and into the cathode (making it negative). … The cathode is an electron donor and can cause reduction to occur. The negatively charged electrode will attract positive ions (cations) toward it from the solution.
What is electrode in chemistry?
An electrode by definition is a point where current enters and leaves the electrolyte. … Electrodes are vital components of electrochemical cells. They transport produced electrons from one half-cell to another, which produce an electrical charge.
What is an anode on a boat?
Boat anodes are an essential part of the boat maintenance cycle as they protect underwater metals from galvanic corrosion. Also known as sacrificial anodes, boat anodes generally come in three metals – aluminium, magnesium and zinc.
What is meant by cell constant?
The cell constant is defined as the ratio of distance between the electrodes which is divided by the area of the cross-sectional of the electrode or we can say that the cell constant is defined as the ratio of distance between the conductance titration electrodes which is measured from the determination resistance of …
What is anode and cathode in electrolytic cell?
Electrochemical cells have two conductive electrodes, called the anode and the cathode. The anode is defined as the electrode where oxidation occurs. The cathode is the electrode where reduction takes place. … In between these electrodes is the electrolyte, which contains ions that can freely move.
What is anode and cathode in diode?
Diode Polarity & Symbols One side is the positive terminal, called the anode. The other terminal is the negative end, called the cathode. Going back to our flow of electricity, current can only move in a diode from the anode to the cathode, never the other way around.
Why anode is positive in electrolytic cell?
In an electrolytic cell(one used for electrolysis), the anode is positively charged. This is because the electrode connected to the positive terminal of the battery is where the negatively charged ions go to lose electrons, that is, to get oxidized.
Who discovered canal?
An anode ray (also positive ray or canal ray) is a beam of positive ions that is created by certain types of gas-discharge tubes. They were first observed in Crookes tubes during experiments by the German scientist Eugen Goldstein, in 1886.
Who proposed cathode tube?
The earliest version of the CRT was known as the “Braun tube”, invented by the German physicist Ferdinand Braun in 1897. It was a cold-cathode diode, a modification of the Crookes tube with a phosphor-coated screen.
Is cathode ray positive or negative?
Cathode rays are a beam of negatively charged electrons traveling from the negative end of an electrode to the positive end within a vacuum, across a potential difference between the electrodes. How Do the Cathode Rays Work? The cathode is a negative electrode, Anode is the positive electrode.
Is Salt a bridge?
A salt bridge refers to a device used to form an electrochemical cell by providing a means to support the free flow of ions between the oxidation and reduction half-cell components. A salt bridge facilitates corrosion because corrosive reactions typically occur in the presence of electrochemical cells.
What is charge of anion?
What is an anion? An anion has more electrons than protons, consequently giving it a net negative charge.
What is anode and anion?
Anode = Electrode with Positive charge. while, Cation = Radical/Ion having a Positive charge. Anion = Radical/Ion having a Negative charge.