What happens when an egg is placed in a hypotonic solution

Andrew Mitchell
Published Apr 22, 2026
Water is known as hypotonic, ie. … There is a much higher water concentration in the egg than in the syrup so water will pass in the opposite direction. This means that the egg will shrink in size.
What will happen to the egg in a hypotonic solution Why?
In the case of the hypotonic solution, there were more solutes in the egg than in the pure water. So, water flowed into the egg, and as a result, it grew in size.
What happens when something is placed in a hypotonic solution?
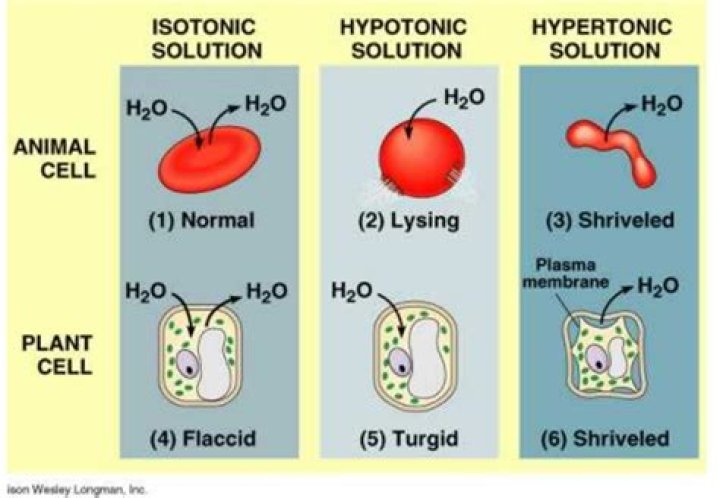
Hypotonic solutions have more water than a cell. Tapwater and pure water are hypotonic. A single animal cell ( like a red blood cell) placed in a hypotonic solution will fill up with water and then burst. This is why putting water on a bloodstained piece of clothing makes the stain worse.
At what point was the egg in a hypotonic solution?
If we put the egg in 95% corn syrup and 5% water then it will be a hypertonic solution, meaning the egg will shrink because the water will be leaving the egg. If we put the egg in 100% water then it will be hypotonic solution, meaning the egg will increase in mass because the water will be entering the egg.What happens when Deshelled egg is placed in hypotonic solution?
Now the egg is covered with only semipermeable membrane. When this is placed in a salt solution (hypertonic solution), exosmosis occurs and the egg shrinks due to loss of water to the surrounding. … if placed in hypotonic solution then the greatest concentration of water is outside the cell. I.e. the cell will swell.
How do each of the three eggs placed in solution compare to the control egg?
How do each of the three eggs placed in solution compare to the control egg? The size of each of the three eggs changed after being placed in the solution. It seems that the eggs that were placed in the 100% corn syrup and 10% corn syrup/ 90% distilled water decreased in size, but the volume of the solution increased.
What happens when you put an egg in water for 24 hours?
Leave the egg in the water for 24 hours. The water will migrate from the side of the membrane where water molecules are abundant (outside the egg) to the side where water molecules are less abundant. After 24 hours, the egg will be plump again.
What happens if you put an egg in vinegar for 24 hours?
If you soak an egg in vinegar the eggshell will absorb the acid and break down, or dissolve. The calcium carbonate will become carbon dioxide gas, which will go into the air. … Soak one egg in vinegar for 24 hours (1 day), one egg for 48 hours (2 days) and one egg for 72 hours (3 days).How do egg membranes dissolve?
Place the egg in the glass and submerge with vinegar. Pour enough vinegar (or cola) over the egg until it is completely submerged in the liquid. The chemical reaction between the acetic acid in the cola and the calcium carbonate in the eggshell causes the shell to dissolve.
What is egg osmosis?In one day the shell will be gone and water will begin moving into the egg. Osmosis is the movement of water through a semi-permeable membrane from an area of higher water concentration (the vinegar) to an area of lower water concentration (the egg).
Article first time published onWhy do cells burst in a hypotonic solution?
Cells placed in a hypotonic solution will take in water across their membrane until both the external solution and the cytosol are isotonic. … If placed in a hypotonic solution, water molecules will enter the cell, causing it to swell and burst.
Why do cells swell up in hypotonic solution?
If the cell is placed in a hypotonic solution, the water potential of the surrounding medium will be higher as compared to the water potential inside the cell. Thus, water will enter into the cell through osmosis and the cell will become swollen.
What causes osmosis?
Osmosis occurs according to the concentration gradient of water across the membrane, which is inversely proportional to the concentration of solutes. … Osmosis occurs when there is a concentration gradient of a solute within a solution, but the membrane does not allow diffusion of the solute.
When a de shelled egg is placed in a concentrated salt solution the egg shrinks Why?
A de shelled egg is surrounded by a semi permeable membrane. When placed in a concentrated salt solution for five minutes, water will flow from the cell to the solution that is exosmosis will take place causing the egg to shrink. This happens because salt solution is hypertonic.
When a boiled egg with its shell removed is placed in water the egg?
The shell of an egg is mainly made up of calcium bicarbonate. It is impermeable to water. So, when the shelled egg is placed in water, the water can neither enter nor leave the egg. Hence, there will not be any change in the size of the egg.
What will happen to a shelled raw egg and Deshelled boiled egg when these are placed in water?
There would be no difference in the eggs as 1. if it is a shelled raw egg then water neither can enter nor leave the egg due the presence of the non permeable hard shell. … If the egg is boiled and deshelled still it won’t make difference because the egg is cooked and then its contents have turned to solid.
What happens when an egg is placed in water?
When you put the egg into pure water, some of the water molecules from the solution move into the egg and the egg swells up. … This happens because, while most of the water molecules flow out of the egg, after some time passes, water molecules pass into the egg at the same rate as they are flowing out.
What happens if you put an egg in water for 48 hours?
Leave the egg in the water for 24 hours. Osmosis will occur; that is, the water will migrate from the side of the membrane where water molecules are abundant (i.e. outside the egg) to the side where water molecules are less abundant (inside the egg). After 24 hours, the egg will be plump again!
When the egg was placed in the water in which direction did?
1. When the egg was placed in the water, in which direction did the water molecules move? The water molecules moved into the egg.
Which solution was hypertonic to the egg?
A concentrated sugar solution is hypertonic compared to the solution inside the egg. Therefore when the egg is placed in the concentrated sugar solution water flows out of the egg, causing it to shrivel.
What is in hypertonic solution?
Hypertonic solution: A solution that contains more dissolved particles (such as salt and other electrolytes) than is found in normal cells and blood.
Why don t red blood cells swell or shrink in blood?
Red blood cells don’t swell or shrink in blood because blood is an isotonic solution compared to the cytoplasm in the red blood cells.
What happens when you put an egg in salt water?
Saltwater is denser than fresh water because of its salt content. The egg will sink in the fresh water because it has greater density than the water. The egg will float in the salt water because when salt is added to water its density becomes greater than that of the egg. That makes the egg float.
What is the conclusion of the bouncy egg experiment?
Conclusions. The eggshell is dissolved because vinegar is an acid and eggshells contain calcium carbonate, which is a base. When these two chemicals are combined, a chemical reaction occurs. Carbon dioxide is formed, which is why you see the bubbles.
What happens to an egg in soda?
If you leave the egg in the soda long enough (a few days), the entire shell will dissolve. Kids often have fun with the egg sac that’s left – it’s like a bouncy water balloon! This helps keep the experiment fun and allows the kids a chance to interact with the eggs.
Why does an egg in vinegar become bouncy?
The Science Behind It: The shell of an egg is made of calcium carbonate! When the egg is placed into the vinegar, you see bubbles, which is the chemical reaction of the acid within the vinegar reacting with the calcium carbonate of the egg shell to produce carbon dioxide.
How do you make a bouncy egg that won't break?
- Place an egg in a cup. Place a raw egg carefully in a clear jar or cup.
- Add vinegar. Pour enough vinegar in the cup to completely submerge the egg.
- Add food coloring. Add in a few generous drops of food coloring and stir gently. …
- Wait! Observe the eggs each day. …
- Rub off the shell. …
- Bounce it!
How long does it take for an egg to turn into rubber?
Most eggs will be rubbery in one to two days, but some eggs may require a third day.
What happens when you put an egg in milk?
Milk won’t make eggs creamier, fluffier, or stretch the dish out. What the milk really does is dilute the flavor of the eggs, making them rubbery, colorless, and something similar to what you would find at a school cafeteria.
How does an egg represent a cell?
The de-shelled eggs are good models of human cells. After the eggshell is gone, a white membrane remains. (It’s actually two membranes, but they’re held tightly together.) This membrane, like those of human cells, is selectively permeable, and the entire egg can be a model for a single cell.
What's it called when a cell bursts?
Cytolysis, or osmotic lysis, occurs when a cell bursts due to an osmotic imbalance that has caused excess water to diffuse into the cell. … The presence of a cell wall prevents the membrane from bursting, so cytolysis only occurs in animal and protozoa cells which do not have cell walls.