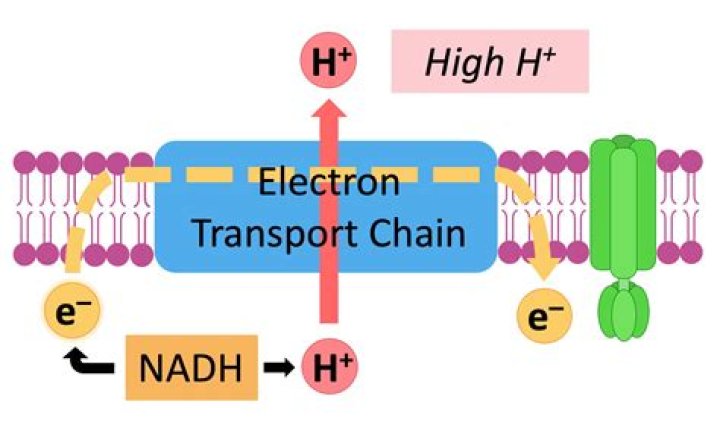
What happens to an electron during luminescence

Zoe Patterson
Published Apr 18, 2026
Luminescence describes the occurrence of luminous effects that are caused by the change of an electron from an excited state to a state with lower energy. Electrons can exist in different energy states. … If electrons absorb energy, they can be elevated to a higher energy level, an excited state.
What happens in luminescence?
Luminescence emission occurs after an appropriate material has absorbed energy from a source such as ultraviolet or X-ray radiation, electron beams, chemical reactions, and so on. … The excitation involves only the outermost electrons orbiting around the nuclei of the atoms.
What is the mechanism of luminescence?
The luminescent process consists of the excitation of an electron to the metastable level, the activated release of the electron to the emitting level, and a forbidden transition between the emitting and the ground state. With high excitation energy, the metastable state is bypassed.
What happens in an atom when light is produced by luminescence?
Luminescent light is produced when, only the electrons of an atom, as opposed to the entire atom, absorb energy and release it as electromagnetic radiation, usually as light. … Electrons absorb the energy supplied to them and jump from a low energy state called, ground state, to a higher energy level.What affects luminescence?
Luminescence of any substance is strongly affected by temperature. Luminescence changes its appearance with temperature variations and, generally, lessens in intensity with an increase in temperature until it eventually vanishes.
Can a person be luminescent?
According to a study conducted in 2009 by Japanese researchers, human bioluminescence in visible light exists – it’s just too dim for our weak eyes to pick up on. “The human body literally glimmers,” the team from the Tohoku Institute of Technology wrote in their study published in PLOS One.
What is electron luminescence?
Electroluminescence (EL) is a process by which photons are generated when the excess electron–hole pairs are created by an electric current caused by an externally applied bias. From: Characterization of Semiconductor Heterostructures and Nanostructures (Second Edition), 2013.
What happens when electrons return to their ground state?
An electron in an excited state can release energy and ‘fall’ to a lower state. When it does, the electron releases a photon of electromagnetic energy. … When the electron returns to the ground state, it can no longer release energy but can absorb quanta of energy and move up to excitation states (higher orbitals).What causes electrons to become excited?
When an electron temporarily occupies an energy state greater than its ground state, it is in an excited state. An electron can become excited if it is given extra energy, such as if it absorbs a photon, or packet of light, or collides with a nearby atom or particle.
When the luminescence is activated by heat then it is called?Fluorescence. —Luminescence that stops within 10–5 seconds after the energy source is removed. Incandescence. —Light created by heating.
Article first time published onIs light released during excitation?
The excitation energy is then emitted as infrared photons, not as visible light, so that luminescence is quenched. These ions, therefore, are called killers—the killing process being opposite to stimulation.
When excitation of electrons by chemical What is the phenomenon called?
Fluorescence is an underlying optical phenomenon that is commonly used in biosensors. Fluorescence occurs when a fluorescent atom absorbs a photon of high energy which excites an electron into one of the many higher energy states.
What is luminescence briefly explain three types of luminescence?
Luminescence is the emission of light produced by methods other than heat. Luminescence is caused by the movement of electrons into different energetic states. There are many different types of luminescence including bioluminescence, chemiluminescence, phosphorescence, and fluorescence.
What is luminescence effect?
Luminescence is spontaneous emission of light by a substance not resulting from heat; or “cold light”. It is thus a form of cold-body radiation. It can be caused by chemical reactions, electrical energy, subatomic motions or stress on a crystal.
What is luminescence process?
Luminescence is a general term to describe the process in which a material absorbs energy from an external source and re-emits that energy in the form of visible light.
What color is luminescence?
It is made from phosphors such as silver-activated zinc sulfide or doped strontium aluminate, and typically glows a pale green to greenish-blue color. The mechanism for producing light is similar to that of fluorescent paint, but the emission of visible light persists long after it has been exposed to light.
What is luminescence decay?
luminescence decays are systematically observed, with short and long lifetimes around 45ps. and 250ps. This behavior is attributed to the band edge exciton fine structure with a dark level. lying a few meV below a bright one.
How do electroluminescent night lights work?
Electroluminescent (EL) lighting are luminaires that use electroluminescence to produce illumination. These devices emit light when a current passes through a thin layer of phosphor or semiconductor, or a strong electromagnetic field is applied.
What is led and how it works?
LED stands for light emitting diode. LED lighting products produce light up to 90% more efficiently than incandescent light bulbs. How do they work? An electrical current passes through a microchip, which illuminates the tiny light sources we call LEDs and the result is visible light.
What animals can see humans glow?
Bioluminescent creatures such as glowworms, fireflies, and plankton are a popular subject with photographers with the low-light abilities of modern digital cameras. The human body’s bioluminescence is 1,000 times less intense than the sensitivity of the human eye, but perhaps in the (distant?)
What is a luminous person?
Someone or something luminous is bright and shiny, or clear and wonderful in a way that reminds you of a bright light.
Is the moon a luminescent?
No, the moon is not a luminous object. Moon has no light of its own.
What is released when an electron loses energy?
Electromagnetic radiation in the form of light is released when an electron loses energy.
Why do electrons not lose energy?
The electrons radiate only when jumping from one orbit to another one. … 1) If an electron is in the electric field of a nucleus, the electron can occupy only certain energy levels. When it is sitting on one of these energy levels, it does not radiate, it does not loose energy.
What happens when an electron jumps from higher to lower?
For example, if an electron jumps from a higher to a lower energy level, the lost energy will have to go somewhere and in fact will be emitted by the atom in a bundle of electromagnetic radiation. … One common way is for the atom to absorb a photon of just the right frequency.
What happens when electrons change levels?
When the electron changes levels, it decreases energy and the atom emits photons. The photon is emitted with the electron moving from a higher energy level to a lower energy level. The energy of the photon is the exact energy that is lost by the electron moving to its lower energy level.
How does an electron absorb energy and re emit it as light?
The electron can gain the energy it needs by absorbing light. If the electron jumps from the second energy level down to the first energy level, it must give off some energy by emitting light. The atom absorbs or emits light in discrete packets called photons, and each photon has a definite energy.
What is difference between bioluminescence and luminescence?
As nouns the difference between bioluminescence and luminescence. is that bioluminescence is (biology|biochemistry) the emission of light by a living organism (such as a firefly) while luminescence is (physics) any emission of light that cannot be attributed merely to the temperature of the emitting body.
What happens when a substance absorbs radiation?
In physics, absorption of electromagnetic radiation is how matter (typically electrons bound in atoms) takes up a photon’s energy — and so transforms electromagnetic energy into internal energy of the absorber (for example, thermal energy).
What wavelength is luminescence?
The excitation spectra are different from the absorption spectra and all the samples have absorption extended to 200 nm or even shorter. So, the excitation at 200–300 nm range can produce luminescence.
Can electrons be excited by heat?
Yes, every material electrons is excited by heat but as the material change heat requirements will also get changed to break the bond between electrons and protons. Conductor: At the temperature ~1degree Celsius most of the conductor electrons get the amount of heat energy to break the bond.