What effect do the polarity size and shape of a molecule have on its physical properties

Mia Walsh
Published Apr 22, 2026
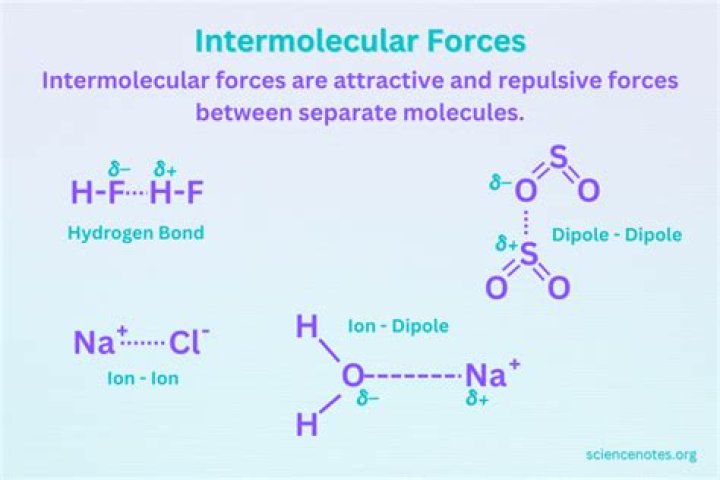
The polarity of a molecule has a strong effect on its physical properties. Molecules which are more polar have stronger intermolecular forces between them, and have, in general, higher boiling points (as well as other different physical properties).
What effect do the size of a molecule have on its physical properties?
The size of the melting or boiling point will depend on the strength of the intermolecular forces. The presence of hydrogen bonding will lift the melting and boiling points. The larger the molecule the more van der Waals attractions are possible – and those will also need more energy to break.
How does the size of a molecule affect polarity?
Polar molecules can bond with each other via dipole-dipole interactions, which are generally stronger than van der Waals forces. Thus, if two molecules are similar in size and one is polar while the other is non-polar, the polar molecule will have higher melting and boiling points compared to non-polar molecule.
How does the shape of a molecule affect its polarity?
The shape of the molecule will determine the direction of each of the individual bond dipoles, and thus, will always play a role in determining the polarity of the molecule as a whole.How does shape affect physical properties?
The very nature of a molecule is dependent upon the shape as well as the electron shift in bonding. The shape of a molecule influences the physical and chemical properties of the elements involved. Determining the shape of a molecule is essential to understanding molecules in chemical reactions.
What is affected by the size of the molecule?
Larger molecules will also diffuse faster when the concentration gradient is steeper. For example, if the concentration of a molecule is much higher inside the cell than outside, the molecules will leave the cell more quickly than when the concentration difference between the two regions is minimal.
How does the shape of a molecule affect its function?
Molecular shape is crucial in biology because of the way it determines how most molecules recognize and respond to each other. One nerve cell in the brain signals another by releasing molecules of a specific shape to go find matching receptor molecules on the surface of the receiving cell.
What would cause the shape of a molecule to be tetrahedral?
Explanation: If there are 4 electron pairs around a central atom, the shape, the geometry that results in LEAST electrostatic interaction is the tetrahedron. For methane, CH4 , the 8 electrons that comprise the 4 C−H bonds demonstrably tend to adopt a shape that minimizes electrostatic repulsion.Which affects the shape of a molecule?
The shape of a molecule is determined by the location of the nuclei and its electrons. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. Thus, the molecule’s shape reflects its equilibrium state in which it has the lowest possible energy in the system.
How does a water molecules shape affect its polarity?Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule.
Article first time published onHow does the polarity of molecule affects solubility?
Polarity. … Polarity plays a pivotal role in solubility. A polar solute will dissolve in a polar solvent whereas a non-polar solvent will dissolve in a non-polar solvent. If we put a polar solute in a non-polar solvent, it will not dissolve.
Does the shape of molecule matter why?
Molecular shape (the shape that a single molecule has) is important in determining how the molecule interacts and reacts with other molecules. Molecular shape also influences the boiling point and melting point of molecules. If all molecules were linear then life as we know it would not exist.
Why does polarity have an effect on the strength of attraction between molecules?
Polar molecules align so that the positive end of one molecule interacts with the negative end of another molecule. Unlike covalent bonds between atoms within a molecule ( intramolecular bonding), dipole-dipole interactions create attractions between molecules of a substance ( intermolecular attractions).
How does polarity affect physical properties?
The polarity of a molecule has a strong effect on its physical properties. Molecules which are more polar have stronger intermolecular forces between them, and have, in general, higher boiling points (as well as other different physical properties).
How is polarity related to physical properties?
In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end. … Polarity underlies a number of physical properties including surface tension, solubility, and melting and boiling points.
How does molecular geometry affect the properties of molecules?
It is important to be able to predict and understand the molecular structure of a molecule because many of the properties of a substance are determined by its geometry. Examples of these properties include polarity, magnetism, phase, color, and chemical reactivity.
What does the shape of small molecules have to do with the shape of large molecules?
Explaining molecular shapes For large molecules, such as proteins, the overall shape will be due to the bonding between atoms as well as other electrostatic and steric effects. VSEPR theory is used to explain the 3D shape around an atom in a molecule and for small molecules this can give the overall shape.
How does shape determine function?
Explanation: A protein’s shape is determined by the sequence of amino acids that make up the protein. The order of the amino acids, or the primary structure, determines the folding of the amino acid chains or tertiary structure. … A protein’s shape determines it function.
Why does the size of a molecule affect diffusion?
Mass of Particle: Heavier particles will move more slowly and so will have a slower rate of diffusion. Smaller particles on the other hand will diffuse faster because they can move faster.
Does polarity affect the rate of diffusion?
Polarity does affect diffusion but only in the case where diffusion is occurring across the cell membrane.
How does size of molecule affect diffusion into cells?
Mass of the molecules diffusing: Heavier molecules move more slowly; therefore, they diffuse more slowly. The reverse is true for lighter molecules. Temperature: Higher temperatures increase the energy and therefore the movement of the molecules, increasing the rate of diffusion.
What determines the polarity of a molecule?
The terms “polar” and “nonpolar” usually refer to covalent bonds. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; if the result is between 0.4 and 1.7, then, generally, the bond is polar covalent.
What electrons affect the shape of the molecule?
The valence shell electron pair repulsion (VSEPR) model predicts the shape of individual molecules based on the extent of electron-pair electrostatic repulsion. … This means that the bonding (and non-bonding) electrons will repel each other as far away as geometrically possible.
Which affects the shape of a molecule quizlet?
The lone pair on the central atom. The lone pair pushes the electron pairs in bonds away, forcing the molecule into a different shape.
Are tetrahedral molecules polar?
A tetrahedral shape is a nonpolar shape: the terminal atoms are distributed in a way that the molecule has no imbalance in polarity based on the shape alone.
What is a tetrahedral shape?
Tetrahedral. Tetra- signifies four, and -hedral relates to a face of a solid; “tetrahedral” literally means “having four faces. ” This shape is found when there are four bonds all on one central atom, with no lone electron pairs. In accordance with the VSEPR theory, the bond angles between the electron bonds are 109.5o …
How tetrahedral voids are formed?
The atom in the tetrahedral void is in contact with four atoms placed at four corners of a tetrahedron. This void is formed when a triangular void made coplanar atoms (first layer) is in contact with the fourth atom above or below it (second layer).
What affects the shape of a water molecule?
The water molecule is bent in shape because the lone pairs of electrons take up space on one side of the molecule, with the hydrogen atoms on the other side, and polar because oxygen’s electronegativity draws electrons strongly to itself, creating a strong overall dipole.
What if water molecules has a linear shape?
The bonds between hydrogen and oxygen are polar because oxygen is more electronegative than hydrogen. Water also exhibits the second kind of polarity. … If the molecule were linear rather than bent, the electron distribution would be even, not uneven, so the molecule would NOT be polar.
Why does polarity increase solubility?
Notice that solubility increases with increasing molecular mass or polarity. … As a result of favorable dipole-dipole attractions between solvent molecules and solute molecules, polar liquids tend to dissolve readily in polar solvents. Water is not only polar but also able to form hydrogen bonds.
Which is true about polarity and solubility?
Thus, polarity affects solubility. If solute and solvent have approximately the same polarity, they will probably form a solution. “Like dissolves like”: Polar solutes dissolve in polar solvents; nonpolar solutes dissolve in nonpolar solvents.