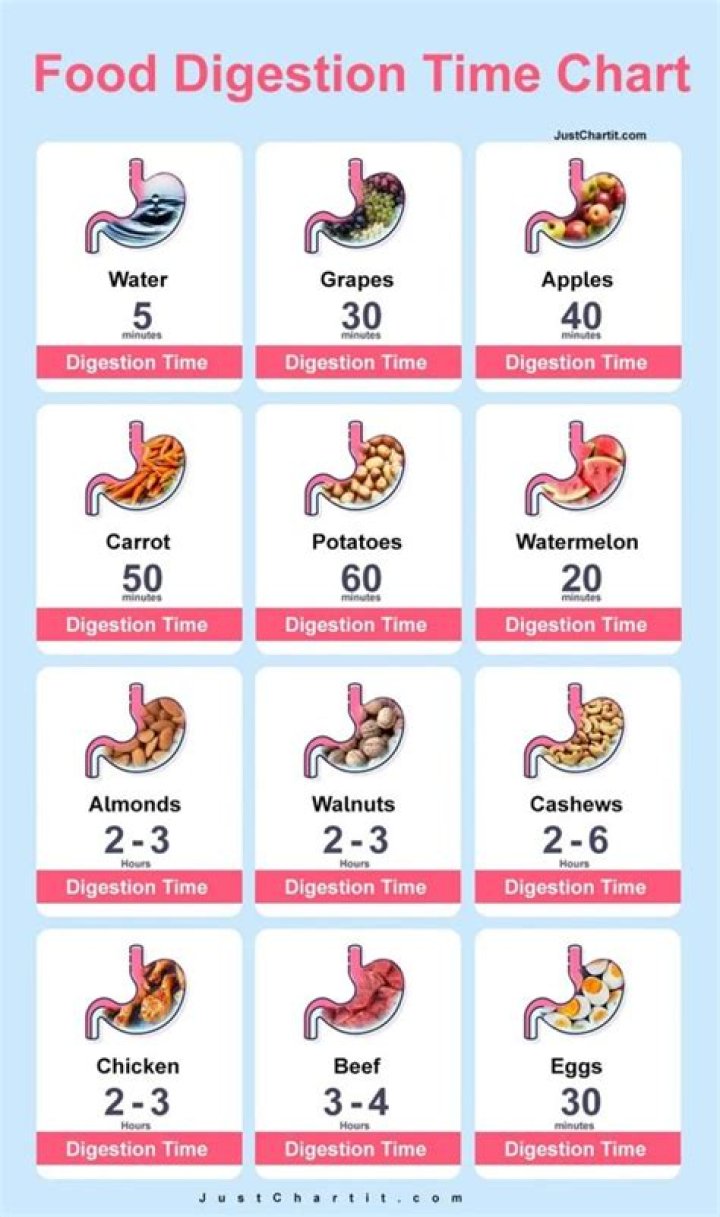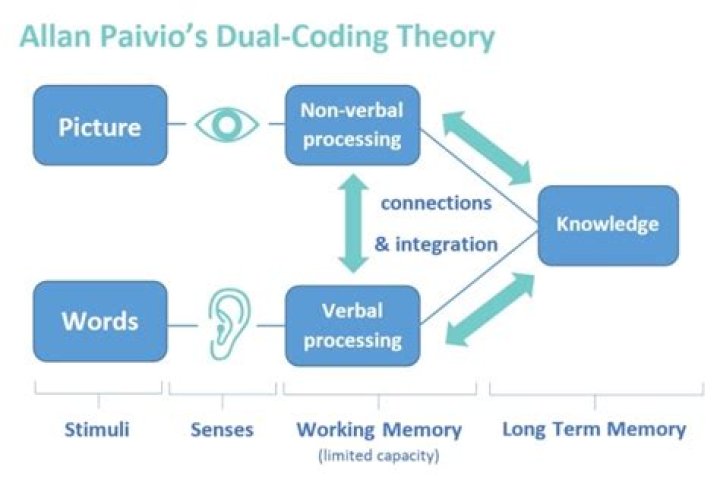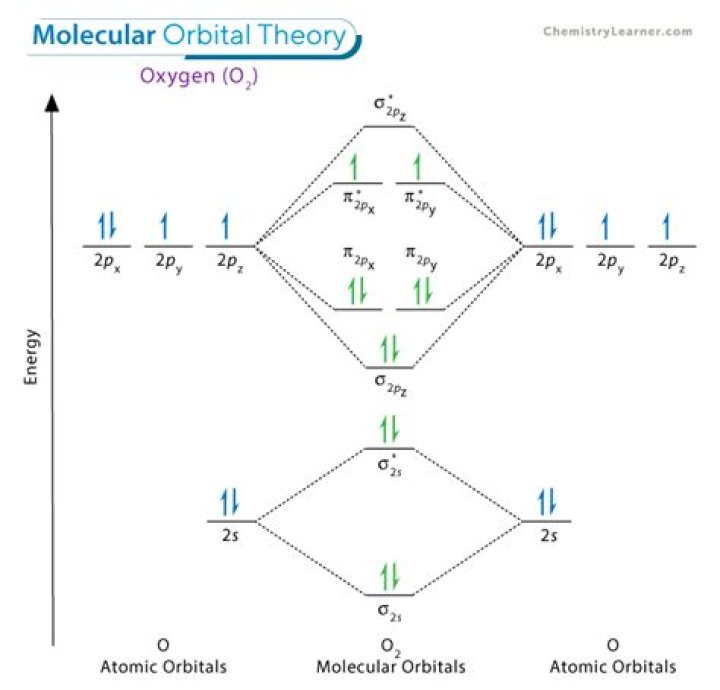
What are the main features of molecular orbital theory

Nathan Sanders
Published Apr 23, 2026
Salient features: (i) Molecular orbitals are formed by the linear combination of atomic orbitals
What are the main features of mot?
Salient Features of the Molecular Orbital Theory The atomic orbitals merge with each other to form the molecular orbital. The electrons of the molecules fill the new energy states of the molecular orbitals, similar to filling up of the energy states of the atomic orbitals.
What is molecular orbital theory class 11?
In simple terms, the molecular orbital theory states that each atom tends to combine together and form molecular orbitals. As a result of such arrangement, electrons are found in various atomic orbitals and they are usually associated with different nuclei.
What is basic postulates of molecular orbital theory?
Postulates of Molecular Orbital Theory: The total number of molecular orbitals formed is equal to the total number of atomic orbital offered by atomic species.The electrons in the molecular orbital are filled in the increasing order of orbital energy (from orbital having lower energy to orbital having higher energy).What is meant by molecular orbital?
A molecular orbital (or MO), in chemistry, refers to an orbital in atomic structure of molecules. It is a wave-function of a molecule’s electron and is used to calculate its chemical and physical properties.
What is the main difference between valence bond theory and molecular orbital theory?
The main difference between valence bond theory and the molecular orbital theory is that valence bond theory explains the hybridization of orbitals whereas the molecular orbital theory does not give details about the hybridization of orbitals.
What are the limitations of molecular orbital theory?
MO theory says that the electrons are delocalized. That means that they are spread out over the entire molecule. The main drawback to our discussion of MO theory is that we are limited to talking about diatomic molecules (molecules that have only two atoms bonded together), or the theory gets very complex.
Why was the molecular orbital theory proposed?
Molecular Orbital (MO) Theory is another theory to explain chemical bonding using orbitals. It was developed about the same time Valence Bond Theory was developed, primarily by Mulliken and Hund. (Mulliken was mentioned earlier because he proposed a definition of electronegativity, and Hund because of Hund’s Rule.)What is the difference between BMO and ABMO?
B.M.O is formed when the lobes of combining atomic orbitals have the same sign. A.B.M.O is formed when the lobes of combing orbitals have the opposite sign. B.M.O is formed by the constructive interference of electron waves. A.B.M.O is formed by the destructive interference of electron waves.
What are the salient features of valence bond theory?(1) Coordination compounds consist of complex ions, in which ligand coordinates with the metal. (2) The ligand coordinates by donating a pair of electrons to the metal. (3) An adequate number of empty orbitals are made available by the metal atom or ion for the formation of coordinate bonds with suitable ligands.
Article first time published onWhat is difference between atomic and molecular orbital?
The main difference between atomic and molecular orbital is that the electrons in an atomic orbital are influenced by one positive nucleus, while the electrons of a molecular orbital are influenced by the two or more nuclei depending upon the number of atoms in a molecule.
What are the conditions for the formation of molecular orbital from atomic orbitals?
For the formation of the proper molecular orbital, proper energy and orientation are required. For proper energy, the two atomic orbitals should have the same energy of orbitals and for the proper orientation, the atomic orbitals should have proper overlap and the same molecular axis of symmetry.
Can molecular orbital theory predict stability?
We can therefore use a molecular orbital energy-level diagram and the calculated bond order to predict the relative stability of species such as H2+.
What is the importance of valence bond theory?
An important aspect of the valence bond theory is the condition of maximum overlap, which leads to the formation of the strongest possible bonds. This theory is used to explain the covalent bond formation in many molecules.
Is O2 stable according to MO theory?
In O2 and F2, there is a crossover of the sigma and the pi ortbials: the relative energies of the sigma orbitals drop below that of the pi orbitals’. Information from the MO diagram justify O2’s stability and show that it’s bonding order is 2.
What are the properties which are better explained by molecular orbitals than valence bond theory?
Valence Bond (VB) theory and Lewis Structures explain the properties of simple molecules. Molecular Orbital (MO) theory better explains the properties of more complex molecules. MO theory explains the partial bonds of NO₃⁻ without using resonance.
Why is the molecular orbital theory better than valence bond theory?
Unlike the VB theory, which is largely based off of valence electrons, the MO theory describes structure more in depth by taking into consideration, for example, the overlap and energies of the bonding and antibonding electrons residing in a particular molecular orbital.
What is the difference between antibonding and bonding molecular orbital?
The main difference between bonding and antibonding molecular orbitals is that bonding molecular orbitals represent the shape of a molecule whereas antibonding molecular orbitals do not contribute to the determination of the shape of a molecule.
What are the main difference between bonding and antibonding molecular orbital?
Bonding molecular orbitalAntibonding molecular orbitali) It is formed by the addition overlap of atomic orbitals.i) It is formed by subtraction overlap of atomic orbital.ii) It may or may not have a node.ii) It always has a node in between the nuclei of bonded atoms.
What is the difference between Antibonding and nonbonding?
The key difference between antibonding and nonbonding is that antibonding orbitals increase the energy of a molecule whereas nonbonding orbitals do not change the energy of a molecule. The terms antibonding and nonbonding come under the molecular orbital theory.
How does molecular orbital theory explain resonance?
Molecular Orbital theory starts by assuming that the three atomic p orbitals on the O atoms overlap to form three molecular π orbitals that extend over the whole molecule. … Thus, Molecular Orbital theory explains resonance delocalization automatically as the natural state of the molecule.
What is valence bond theory and its limitations?
Limitations of Valence Bond Theory It has its own set of limitations. They are: It fails to explain the tetravalency of carbon. This theory does not discuss the electrons’ energies. The assumptions are about the electrons being localized to specific locations.
What is the highest occupied molecular orbital?
Frontier Orbitals. Frontier orbitals, that is, the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), are important indicators for the effect of adsorption on a metal.
What is the name of the lowest energy empty molecular orbital?
Lowest Unoccupied Molecular Orbital. The LUMO is the lowest energy orbital that has the scope to accept electrons and hence it acts as an electron acceptor and characterizes the susceptibility of the molecule toward attack by nucleophiles.
Do molecular orbitals have the same names as atomic orbitals?
The major difference between atomic and molecular orbitals is that atomic orbitals represent electron density in space associated with a particular atom. Molecular orbitals are associated with the entire molecule, meaning the electron density is delocalized (spread out) over more than one atom.
How do electrons fill the molecular orbital theory?
Electrons fill molecular orbitals following the same rules that apply to filling atomic orbitals; Hund’s rule and the Aufbau principle tell us that lower-energy orbitals will fill first, electrons will spread out before they pair up, and each orbital can hold a maximum of two electrons with opposite spins.
Does molecular orbital theory follow Hund's rule?
Filling Electrons in MO Diagrams Three general rules apply: … Hund’s rule states that when there are several MOs with equal energy, and the electrons occupy the MOs one at a time before two occupy the same MO.
What do you understand by bonding and antibonding molecular orbitals What are the conditions for combination of atomic orbitals?
Placing an electron in the bonding orbital stabilizes the molecule because it is in between the two nuclei. Conversely, placing electrons into the antibonding orbitals will decrease the stability of the molecule. … They will first fill the lower energy orbitals, and then they will fill the higher energy orbitals.
Which molecular orbital diagram is most stable?
The head-to-head overlap giving σ molecular orbitals results in greater overlap, making its bonding molecular orbital the most stable and lowest energy, while the σ* antibonding is least stable and has the highest energy (Figure 9.24 “Molecular orbital energy diagram for homonuclear diatomic molecules made from atoms …
How many electrons can a molecular orbital hold?
We then fill the orbitals with the required number of valence electrons according to the Pauli principle. This means that each molecular orbital can accommodate a maximum of two electrons with opposite spins.