No+ Lewis Structure - How To Discuss

Rachel Ross
Published Apr 29, 2026
No+ Lewis Structure
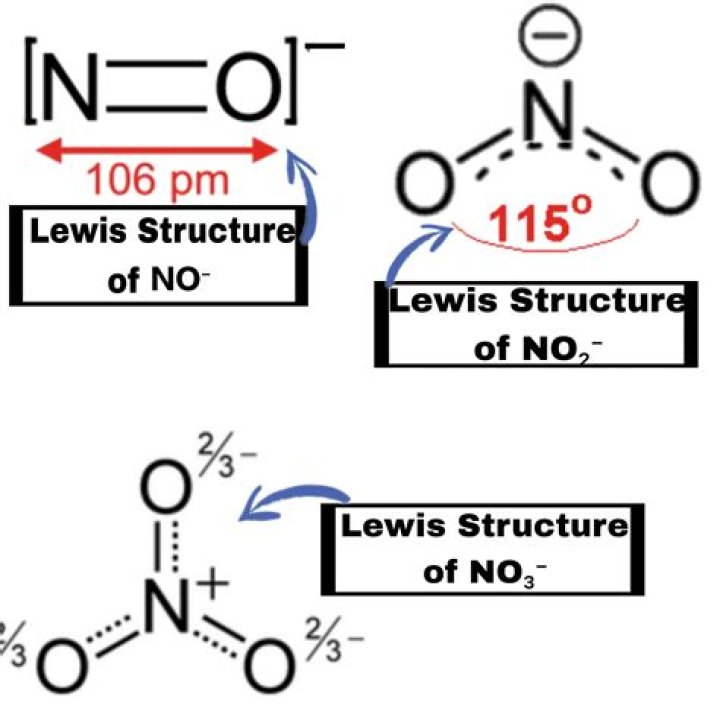
In the best Lewis structure of NO +, what is the regular charge of N? 3
N is 3.
You can only determine this by looking at the ic table.
I don't really understand your picture, but it's about the cost of O and N labor. Nitrogen prefers a covalent bond and 1 solitary pair. Therefore, they can be stable and follow the byte principle. So if O has 3 links it will be +1 (+2 for 4 links) and if O has only 1 link it will be 1. Isolated pair
No+ Lewis Structure
No+ Lewis Structure
In the best Lewis structure for NO +, what is the formal charge of N? 3
I don't really understand your picture, but it's worth working with O and N. Oxygen likes to have 2 covalent bonds and 2 solitary pairs. Nitrogen likes to have 3 covalent bonds and 1 single pair. Therefore, they can be stable and adhere to the byte principle. So if O has 3 links then it will be +1 (+2 for 4 links) and if O has only 1 link then it will be 1. N must have 4 links to +1 and 2 links to have 1 link, isolated couple easier to see