How does entropy affect free energy

Olivia House
Published Apr 22, 2026
The change in the Gibbs free energy of the system that occurs during a reaction is therefore equal to the change in the enthalpy of the system minus the change in the product of the temperature times the entropy of the system. If the reaction is run at constant temperature, this equation can be written as follows.
How does entropy relate to free energy?
Gibbs free energy combines enthalpy and entropy into a single value. Gibbs free energy is the energy associated with a chemical reaction that can do useful work. It equals the enthalpy minus the product of the temperature and entropy of the system. … If ΔG is positive, then the reaction is non-spontaneous.
Does free energy increase when entropy increases?
A process will only happen spontaneously, without added energy, if it increases the entropy of the universe as a whole (or, in the limit of a reversible process, leaves it unchanged) – this is the Second Law of Thermodynamics.
Does entropy change free energy?
Gibbs free energy, denoted G, combines enthalpy and entropy into a single value. The change in free energy, ΔG, is equal to the sum of the enthalpy plus the product of the temperature and entropy of the system.How does entropy affect energy?
Entropy is a measure of randomness or disorder in a system. … The more energy that is lost by a system to its surroundings, the less ordered and more random the system is. Scientists refer to the measure of randomness or disorder within a system as entropy. High entropy means high disorder and low energy (Figure 1).
How does entropy decrease?
When a small amount of heat ΔQ is added to a substance at temperature T, without changing its temperature appreciably, the entropy of the substance changes by ΔS = ΔQ/T. When heat is removed, the entropy decreases, when heat is added the entropy increases.
What does Gibbs free energy tell us?
Gibbs free energy measures the useful work obtainable from a thermodynamic system at a constant temperature and pressure. Just as in mechanics, where potential energy is defined as capacity to do work, similarly different potentials have different meanings.
What does free energy change depend on?
The standard-state free energy of reaction is a measure of how far the standard-state is from equilibrium. Go depends on the temperature of the reaction. As a result, the equilibrium constant must depend on the temperature of the reaction.Why Does entropy increase?
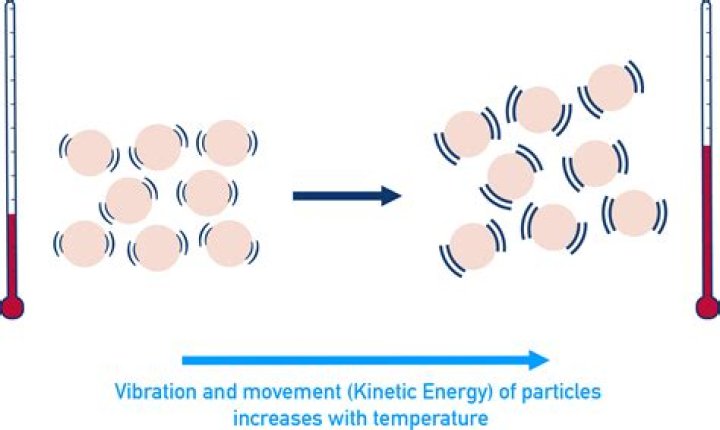
Entropy increases as temperature increases. An increase in temperature means that the particles of the substance have greater kinetic energy. The faster-moving particles have more disorder than particles that are moving slowly at a lower temperature.
How does the entropy change when glucose undergoes combustion?How does entropy change when glucose undergoes combustion in the equation C6H12O6 + 6O2 –> 6CO2 + 6H2O? Entropy increases since the total # of products is greater than the total # of reactants.
Article first time published onWhat happens if entropy decreases?
This is stated by the Entropy Principle. If the entropy of the system decreases, the entropy of the surrounding always increases by a greater value, thus making the entropy of the universe positive.
Why is increasing entropy favorable?
Processes are favorable for happening when the free energy difference is minimized, so an increase in entropy is favorable while a decrease in internal energy is also favorable.
Why free energy is not possible?
Free energy machines do not work. No machine can create energy out of nothing, as this would violate the law of mass-energy conservation, which is fundamental and universal. … Mass can be converted to energy, and energy can be converted to mass, but together they must be conserved.
Does entropy destroy energy?
Energy transforms and dissipates. Entropy is the dispersing state of energy from order to less order. Energy is not destroyed but dispersed or spread out. Hence, entropy increases and energy is conserved, even when dispersed.
What is enthalpy entropy Gibbs free energy?
Enthalpy is the amount of heat energy transferred (heat absorbed or emitted) in a chemical process under constant pressure. Entropy measures the amount of heat dispersed or transferred during a chemical process.
How does entropy increase?
Entropy increases as temperature increases. An increase in temperature means that the particles of the substance have greater kinetic energy. The faster moving particles have more disorder than particles that are moving more slowly at a lower temperature.
How does temperature affect Gibbs free energy?
As temperature increases, -TΔS will become more and more positive, and will eventually outweigh the effect of ΔH. At low temperatures, ΔG will be negative because of the effect of the negative ΔH, but as you increase the temperature, the effect of the positive -TΔS will eventually outweigh that.
What is entropy change?
Entropy change can be defined as the change in the state of disorder of a thermodynamic system that is associated with the conversion of heat or enthalpy into work. A system with a great degree of disorderliness has more entropy.
How does pH affect Gibbs free energy?
The change in Gibbs Free Energy for a reaction ( ΔGrxn) depends on the concentration of reactants and products, so an increase in pH increases ΔGrxn if H3O+ is a reactant, and decreases ΔGrxn if H3O+ is a product.
What affects entropy?
The entropy of a substance increases with its molecular weight and complexity and with temperature. The entropy also increases as the pressure or concentration becomes smaller. Entropies of gases are much larger than those of condensed phases.
How does entropy affect the universe?
Explanation: Energy always flows downhill, and this causes an increase of entropy. Entropy is the spreading out of energy, and energy tends to spread out as much as possible. … The universe will have run down completely, and the entropy of the universe will be as high as it is ever going to get.
Why is entropy important?
Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system. … The concept of entropy provides deep insight into the direction of spontaneous change for many everyday phenomena.
Does entropy increase?
A measure of the level of disorder of a system is entropy, represented by S. … In an irreversible process, entropy always increases, so the change in entropy is positive. The total entropy of the universe is continually increasing.
Is entropy good or bad?
In general entropy is neither good nor bad. There are many things that only happen when entropy increase, and a whole lot of them, including some of the chemical reactions needed to sustain life, would be considered as good.
Does entropy always increase in a spontaneous process?
The second law of thermodynamics states that the total entropy of the universe always increases for a spontaneous process. The net change in entropy of the system, ∆S , equals the sum of the entropy created during the spontaneous process and the change in energy associated with the heat flow.
What change occurs in the free energy during metal corrosion?
Thus, potential is a measure of reaction (corrosion) tendency. If the potential is negative, the metal would be active, that is, corrosion would take place spontaneously because free energy change (ΔG) is negative.
What's the difference between entropy and enthalpy?
Enthalpy is the amount of internal energy contained in a compound whereas entropy is the amount of intrinsic disorder within the compound.
What is the free energy of formation of glucose from carbon dioxide and water?
Heat of formation of glucose from CO2 and H2O = −302,300 cal. One gram of glucose represents −50.7 entropy units. This is the free energy change in glucose formation.
Which statement best explains why water can solvate both negative and positive ions?
Ionic or electrostatic interactions occur between two groups of opposite charge. Hydrophobic or nonpolar substances are insoluble in water. Which statement best explains why water can solvate both negative and positive ions? It is dipolar.
Which of the following types of bonding interactions is not classified as non covalent?
Which of the following types of bonding interactions is NOT classified as non-covalent? Hydrogen bonding.
How are internal energy change free energy change and entropy change are related to one another?
As it happens, enthalpy and entropy changes in a reaction are partly related to each other. The reason for this relationship is that if energy is added to or released from the system, it has to be partitioned into new states. Thus, an enthalpy change can also have an effect on entropy.



