How do you explain conservation of mass

Nathan Sanders
Published Apr 23, 2026
The Law of Conservation of Mass dates from Antoine Lavoisier’s 1789 discovery that mass is neither created nor destroyed in chemical reactions. In other words, the mass of any one element at the beginning of a reaction will equal the mass of that element at the end of the reaction.
How do you explain the law of conservation of mass?
The law of conservation of mass states that in a chemical reaction mass is neither created nor destroyed. For example, the carbon atom in coal becomes carbon dioxide when it is burned. The carbon atom changes from a solid structure to a gas but its mass does not change.
What is conservation of mass short answer?
The law of conservation of mass states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations. According to the law of conservation of mass, the mass of the products in a chemical reaction must equal the mass of the reactants.
How do you explain conservation of mass to a child?
The law of conservation of mass is a fundamental principle of physics. According to this law, matter can be neither created nor destroyed. In other words, the mass of an object or collection of objects never changes, no matter how the parts are rearranged.What is law of conservation of mass explain with example?
The law of conservation of mass states that matter cannot be created or destroyed in a chemical reaction. For example, when wood burns, the mass of the soot, ashes, and gases equals the original mass of the charcoal and the oxygen when it first reacted.
What is conservation of matter 5th grade?
The Law of Conservation of Matter says that the amount of matter stays the same, even when matter changes form. Sometimes it may seem that matter disappears during a science experiment, but this law tells us that matter cannot magically appear or disappear, it simply changes from one form to another.
What is an example of conservation of mass?
The law of conservation of mass states that matter cannot be created or destroyed in a chemical reaction. For example, when wood burns, the mass of the soot, ashes, and gases equals the original mass of the charcoal and the oxygen when it first reacted. So the mass of the product equals the mass of the reactant.
Why is it important to understand the Law of Conservation of Mass?
The law of conservation of mass is very important to the study and production of chemical reactions. If scientists know the quantities and identities of reactants for a particular reaction, they can predict the amounts of products that will be made.How is mass conserved in an ecosystem?
Conservation of Mass Conservation of Matter Over a long time, matter is transferred from one organism to another repeatedly and between organisms and their physical environment. … No matter how substances within a closed system interact, the total mass of the system remains the same.
What is conservation of mass Class 9?Class 9 Chemistry Atoms and Molecules. Laws of conservation of mass. Laws of conservation of mass. The law states that mass can neither be created nor destroyed in a chemical reaction i.e. Total masses of reactants is equal to the sum of masses of products and the masses of unreacted reactants.
Article first time published onWhat is conservation of mass in physics class 11?
The law of conservation of mass states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations. According to the law, the mass of the products in a chemical reaction must equal the mass of the reactants.
What is law of conservation of mass describe with an activity class 9?
The law of conservation of mass states that matter cannot be created nor destroyed, only it’s form changed. Take the example of burning a piece of paper. It seems that the mass shrinks as the paper burns.
How does the conservation of mass relate to this activity?
What is conservation of mass and how does it relate to this exercise? Conservation of mass says the total mass of the reactants must equal the total mass of the products. As long as the carbon dioxide was not allowed to escape, the change in mass that occurred during the reaction should be zero.
Which of the following is a best example of law of conservation of mass?
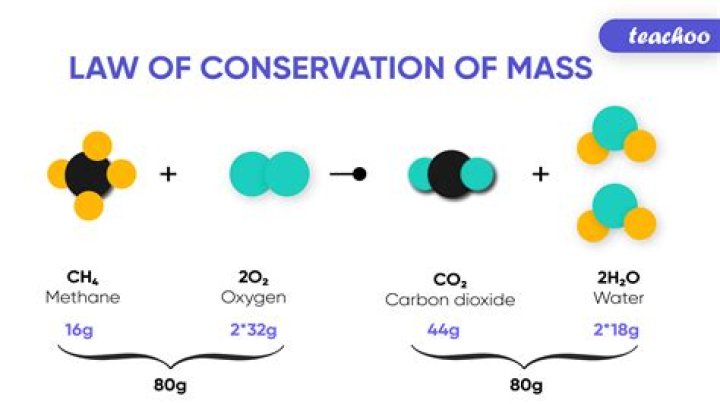
12 g of carbon combines with 32 g of oxygen to form 44 g of CO2 is the best example of law of conservation of mass.
What is law of conservation of mass Class 12?
The law of conservation of mass states that. “The mass in an isolated system can neither be created nor be destroyed but can be transformed from one form to another”. According to the law of conservation of mass, the mass of the reactants must be equal to the mass of the products for a low energy thermodynamic process.
What happened when Dr Jeff boiled the sugar water?
When Dr. Jeff boils the sugar water, what happens? As the water is heated it changes forms from liquid to gas. The sugar is left behind as a solid, providing us with evidence that it was in there the whole time.
What is conservation of matter in science?
Matter is anything that has mass and takes up space. … Matter can change form through physical and chemical changes, but through any of these changes, matter is conserved. The same amount of matter exists before and after the change—none is created or destroyed. This concept is called the Law of Conservation of Mass.
What is law of conservation of mass Class 10?
Law of conservation of mass says that “mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations. According to the law of conservation of mass, the mass of the products in a chemical reaction must equal the mass of the reactants.”
What is the law of conservation of mass quizlet?
The law of conservation of mass states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations.
How does the concept of conservation of mass apply to chemical reactions?
Matter cannot be created or destroyed in chemical reactions. This is the law of conservation of mass. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Balanced chemical equations show that mass is conserved in chemical reactions.
Which of the following statements best explain the law of conservation of mass?
Answer: the law of conservation of mass states that the mass is an isolated system which can not be created nor destroyed .
What is the meaning of conservation law?
conservation law, also called law of conservation, in physics, a principle that states that a certain physical property (i.e., a measurable quantity) does not change in the course of time within an isolated physical system. … In an isolated system the sum of all forms of energy therefore remains constant.
What is the law of conservation of mass Mcq?
The law of conservation of mass states that mass can be neither created nor destroyed in a chemical reaction. Total mass of reactants is equal to total mass of products.
What is conservation of energy class 11?
Law of conservation of energy states that the energy of a system is always constant. In other words, we can say that energy can neither be created nor destroyed. … Mechanical energy (E) is the sum of the potential energy (U) and the kinetic energy (K) of the freely falling body. Therefore, E=K+U=constant.
What is class 11 conserved Ncert?
Conserved quantities are physical quantities that do not change over time. For example, the kinetic and potential energy of a body under external force fluctuate with time, but the total mechanical energy (kinetic + potential) remains constant.
What is conservation of momentum Class 11?
According to the law of conservation of momentum when two bodies collide with one another, the sum of their linear momentum always remains unaffected; that is linear momentum after and linear momentum before the collision remains the same but this is true only when there is no external unbalanced force acting on the …
What is the purpose of a conservation of mass lab?
The purpose of the lab was to discover whether a chemical reaction will cause mass to be created, destroyed, or conserved.