How can we solve the problem of acids and bases

Zoe Patterson
Published Apr 20, 2026
Determine the concentration of the conjugate base.Determine Kb (using Ka if necessary)Write an expression for Kb from the reaction: A1- + H2O <-< HA + OH1-[A1-], and Kb are known: Let [HA] and [OH1-] be X and solve as:Solve for X.
What is a solution in acids and bases?
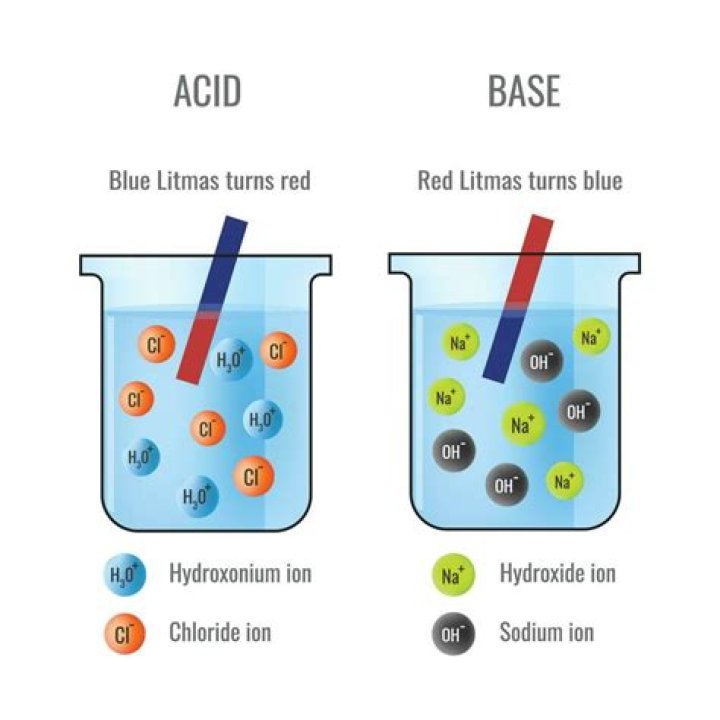
An acidic solution has a high concentration of hydrogen ions (H +start superscript, plus, end superscript), greater than that of pure water. A basic solution has a low H +start superscript, plus, end superscript concentration, less than that of pure water.
How do you neutralize the strong acids and bases?
Strong AcidsStrong BasesHClLiOHHBrNaOHHIKOHHCIO4RbOH
What are the solutions of acids?
Acids are defined by the Bronsted -Lowry definition as compounds that donate a hydrogen ion in solution. Acidic solutions are made by dissolving the acidic compound (as the solute) in water (as the solvent). The pH of acidic solutions is less than 7.How do acids and bases interact in solution?
In an acid-base (or neutralization) reaction, the H+ ions from the acid and the OH- ions from the base react to create water (H2O). Another product of a neutralization reaction is an ionic compound called a salt.
How strong are acid base solutions?
The strength of an acid or base is measured on a scale of numbers called pH scale (0-14). More acidic a solution is lesser will be its pH. Solution with pH=0,1,2,3 are strong acids. Solution with pH=4,5,6 are weak acids.
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
Explanation: Neutralization reaction occurs when a solution of an acid is mixed with a solution of a base in a test tube. The result of the reaction is the production of salt and water with the evolution of heat.
How can an acidic solution be turned into a neutral solution?
When you add a base to this acidic solution, the base accepts protons from the water molecules creating OH− ions. The H3O+ ions and indicator molecule transfer protons to the OH− ions. When enough base is added so that the concentration of H3O+ and OH− ions becomes equal, the solution is neutralized.What are some basic solutions?
Basic solutions contain ions, conduct electricity, turn red litmus paper blue, and feel slippery to the touch. Examples of common basic solutions include soap or detergent dissolved in water or solutions of sodium hydroxide, potassium hydroxide, or sodium carbonate.
When an acid is mixed with a base solution What are the usual end products?When an acid and a base react (combine) releasing equal quantities of H+ ions and OH- ions, neutralization results. H+ ions and OH- ions combine (neutralize each other) to regenerate water. 1. What is an acid?
Article first time published onHow do you neutralize a base spill?
Base spills should be neutralized with boric acid and then cleaned with a paper towel or sponge. Do not use acetic acid or sodium bicarbonate to clean a base spill. The sodium bicarbonate will not neutralize the spill, and acetic acid could react strongly with the base.
How do acids and bases form salts through chemical reactions?
When an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. The H(+) cation of the acid combines with the OH(-) anion of the base to form water. The compound formed by the cation of the base and the anion of the acid is called a salt.
How do you neutralize an acid spill?
Pour baking soda directly onto any acid spill. This will neutralize light acids like vinegar or even strong, dangerous acids like muriatic and sulphuric acids. Douse the entire affected area with the baking soda (sodium bicarbonate, NaHCO3) to neutralize the acid.
How do acid and base react with each other explain with example?
When an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. The H(+) cation of the acid combines with the OH(-) anion of the base to form water. … Here HCl, an acid, reacts with NaOH, a base to give NaCl, salt and water.
How do acids and bases work?
An acid is a substance that donates protons (in the Brønsted-Lowry definition) or accepts a pair of valence electrons to form a bond (in the Lewis definition). A base is a substance that can accept protons or donate a pair of valence electrons to form a bond.
What happens when acid reacts with base explain with example?
When mixed, acids and bases neutralize one another and produce salts, substances with a salty taste and none of the characteristic properties of either acids or bases.
Which of the following statements is correct about an aqueous solution of an acid and of a base?
D – When the pH of an aqueous solution is higher, acidic properties are weak. Thus it is a base. When the pH an aqueous solution is lower, basic properties are weak. Thus it is an acid.
Which of the following gives the correct increasing order of acidic?
The correct increasing order of acidic strength is Water < Acetic acid < Hydrochloric acid.
Which gas is evolved when acids react with metals?
In general, acids react with metals to give salt and release hydrogen gas.
What happens to an acid and base in a water solution?
Acid and bases in water Acids in water solution dissociate H+ ions. Base, when dissolved in water, produces OH– ion. When an acidic solution is diluted with water, the concentration of H+ ions decreases and the pH of the solution increases towards 7.
What is the strength of acids and bases?
CharacteristicStrong Acid or BaseWeak Acid or BaseMoleculesfewlarge numberIonslarge numbersmall numberConductivitystrongweakBond Strengthweakstrong
How do acids and bases react with metals?
In general, acids react with metals and release hydrogen gas. When base reacts with metal it forms salt and release hydrogen gas.
How do you find the base of a solution?
If the pH is lower than 7, the solution is acidic. When pH is higher than 7, the solution is basic. These numbers describe the concentration of hydrogen ions in the solution and increase on a negative logarithmic scale.
How do acids and bases affect the environment?
Acid rain increases acidity in bodies of water like streams and lakes. This alone damages aquatic organisms; however, an increase in acidity also causes aluminum that is found in soil to be leached into bodies of water. The combined impact of aluminum and acidity causes damage to aquatic ecosystems.
What is a base solution in chemistry?
According to the Bronsted-Lowry definition, bases are substances that accept hydrogen ions from an acid. Basic solutions are made by dissolving the base, the solute, in a liquid solvent. Basic solutions are characterized by pH values higher than 7 and can conduct electricity.
Can acids and bases neutralize each other?
Acid and base neutralize each other to form a salt and water.
How are acids bases and neutral solutions related?
A base is a type of chemical that forms solutions that taste bitter due to a low concentration of positive hydrogen ions. Acids will react with bases to form salts. … Neutralization reactions are where an acid and a base react and create a product that is neutral. When they react, they form neutral water and a salt.
How do acids and bases change the pH of a solution?
Acids are substances that provide hydrogen ions (H+) and lower pH, whereas bases provide hydroxide ions (OH–) and raise pH. The stronger the acid, the more readily it donates H+. For example, hydrochloric acid and lemon juice are very acidic and readily give up H+ when added to water.
What happens when you mix acid and base together?
If we mix equal amounts of an acid and a base then two chemicals essentially cancel out each other and produce salt and water. Mixing equal amounts of a strong acid with strong base results in a neutral solution whose pH value remains 7 and this type of reactions are known as neutralization reactions.
What happens if you mix acid and alkali together?
When you add an alkali to an acid a chemical reaction occurs and a new substance is made. If exactly the right amounts of acid and alkali are mixed, you will end up with a neutral solution. This is called a neutralisation reaction.
How do you remove acid from skin?
Some acid burns are made worse if rinsed (flushed) with water. Carbolic acid or phenol does not mix with water, so use alcohol first to flush the chemical off the skin and then flush with water. If alcohol is not available, flush with a large amount of water. Do not flush the eye with alcohol.



