C2h6 O2 Co2 H2o - How To Discuss

Samuel Coleman
Published May 12, 2026
C2h6 O2 Co2 H2o
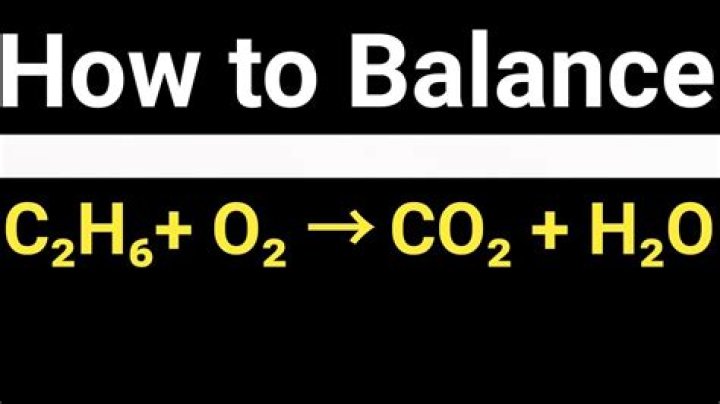
Please balance this C2H6 + O2> CO2 + H20? 3
Thank you very much for your help
I use H2O, not H2O.
We need 3H2O to get 6H.
We need 2CO2 to get 2C.
7O to the right, ie 3.5 O2 to the right. So put everything
2C2H6 + 7O2> 4CO2 + 6H2O
C2h6 O2 Co2 H2o
C2h6 O2 Co2 H2o
C2h6 O2 Co2 H2o
2 C2H6 + 7 O2 = 4 CO2 + 6 H2O
Really needed:
First, separate all the elements and show that there are many elements!
Left (my left) or sideway C2H6 + O2
C2 is H6 and O2.
On the other hand, CO2 + H2O
C1 O3 and H2
2C2H6 + 7O2
C4 and H12 and O14.
> 4CO2 + 6H2O
Manufacturing
C4 and O14 and H12
You still want to multiply the number you added to the item, so you want to make sure it's the same on both sides.
Combustion is the equation
Welcome! = vv = Star Angel Header Moore
C2h6 O2 Co2 H2o
C2h6 O2 Co2 H2o
Do i have But for oxygen, it gives me different reactions, as if they are out of balance.



